The Antidepressant -like Effects of Delta-Opioid Receptor Agonists
Abstract
Activation of the delta-opioid receptor (DOR) system produces an interesting behavioral profile distinct from that of other opioids. Unlike mu- and kappa-opioid agonists, delta-opioid agonists alone have limited pain-relieving qualities as measured in morphine-sensitive antinociceptive assays. Recent evidence, however, suggests that the DOR system may play a role in regulating mood and emotional states. For example, DOR activation stimulates robust antidepressant-like effects in preclinical assays, suggesting that these compounds may have therapeutic potential for treating human depression. This review discusses the role of the DORs in depression and the antidepressant-like effects of delta-opioid agonists as well as their limitations.
Introduction
Following the discovery of endogenous opioids and multiple opioid receptors in the 1970s, the delta-opioid receptor (DOR) was identified as having high affinity for the endogenous opioid peptides met- and leuenkephalin, using isolated tissue assays such as the mouse isolated vas deferens. Instability and low bioavailability of delta peptides slowed the preclinical evaluation of DOR agonists; however, the development of systemically active, selective nonpeptidic delta-opioid agonists has accelerated investigations into the physiological and behavioral changes produced by DOR activation. Initially, it was expected that these compounds would have antinociceptive properties similar to those of other opioids, without the negative side effects such as respiratory depression, physical dependence, and abuse potential. Surprisingly, these nonpeptidic delta-opioid agonists alone had minimal pain-relieving qualities as measured in morphine-sensitive antinociceptive assays, and they also lacked other familiar opioid characteristics like respiratory depression, reinforcing effects as measured in self-administration studies, and opioid (mu or kappa) withdrawal symptoms. Despite these studies, the true nature of the physiological or behavioral effects mediated by DORs has unraveled slowly.
Recent studies have demonstrated that DOR agonists have therapeutic potential as: 1) analgesics under certain conditions, 2) anti-anxiety medications, and 3) treatments for Parkinson Disease. In addition, a number of studies have suggested that DOR activation also produces antidepressant-like effects in preclinical models and in assays that evaluate antidepressant actions of drugs. This manuscript will review the current literature and theories addressing the antidepressant action of DOR agonists and DOR activation.
Depression And Current Treatments
Depression, a mental disorder that affects more than 18 million Americans of all ages each year, is most often characterized by a sad or blunted mood (i.e., affect) or loss of interest or pleasure that coexists with feelings of hopelessness, pessimism, worthlessness, or helplessness. Along with these basic symptoms, other disease characteristics include changes in appetite, constant lethargic state or fatigue, restlessness, irritability, thoughts of death or suicide, as well as chronic pain. Selective serotonin reuptake inhibitors (SSRIs), such as fluoxetine (Prozac®) and sertraline (Zoloft®), are some of the main treatment options used; however, approximately 70% of patients are not adequately treated with an SSRI alone [see http://www.nimh.nih.gov/press/stard.cfm for recent updates about the Sequenced Treatment Alternatives to Relieve Depression (STAR*D), the largest clinical trial for depression treatment in the US]. Therefore, research into new and alternative medications and treatment approaches should be evaluated for therapeutic potential.
The typical antidepressant treatments increase neurotransmitter signaling by preventing reuptake into presynaptic terminals. The most frequently prescribed and used antidepressants are SSRIs, but other drugs are used alone or in combination with SSRIs for treatment. Alternative medications used to treat depression act as reuptake blockers at norepinephrine [amitriptyline (Elavil®)], dopamine [bupropion (Wellbutrin®)], or multiple neurotransmitters such as norepinephrine and serotonin [venlafaxine (Effexor®)] terminals to increase signaling of these neurotransmitters. An alternative approach to treating depression—and one of the most successful treatments available—is electroconvulsive therapy (ECT), which is mainly used for treatment-resistant and severe depression. Some additional non-pharmacological treatments in development are transcranial magnetic stimulation and vagus nerve stimulation (1). Alternative drug targets for this disease are being investigated to find interventions with increased efficacy, faster therapeutic onset, and fewer side effects.
Recently, the opioid system has been proposed as a novel target system for the treatment of depression. Opioids are known to alter mood states; for example, muopioid receptor activation produces euphoria, whereas withdrawal from prolonged opiate use can induce depressive-like symptoms. Some studies have shown that concentrations of the endogenous opioid peptide β-endorphin were decreased in depressed patients as compared with those found in control patients and that these decreased amounts were returned to normal levels following treatment with fluvoxamine (2). Exogenously administered β-endorphin also had antidepressant properties in depressed patients (3–6). Likewise, some opioid ligands, such as cyclazocine, buprenorphine, oxycodone, and oxymorphone, were suggested to have antidepressant actions on their own in humans with refractory or treatment-resistant depression (7–10). In addition, typical antidepressants might produce antinociceptive and antidepressant effects through modulation of endogenous opioids or through gradual changes in opioid receptor expression (2, 11–13). Similarly, changes in opioid signaling have been associated with the antidepressant actions of ECT (14, 15). Despite these data, there is little clinical information specifically linking the DOR with depression or antidepressant drug actions.
Antidepressant Action And The Delta-Opioid Receptor (DOR) System
The association between the DOR system and depression as well as the antidepressant-like effects of opioid ligands are being evaluated in preclinical assays. There is some, limited evidence that the DOR may play a role in depressive states. For example, Filliol et al. demonstrated that DOR-null mice exhibit depressive-like behaviors (16). Alternatively, administration of opioid peptides and DOR agonists has been demonstrated to have antidepressant actions in animal models of depression. For example, exogenously administered enkephalins and endorphins demonstrated antidepressant-like effects in the forced swim test (17) and in the learned helplessness paradigm in rats (18). Likewise, the delta-opioid peptides (Tyr-d-Ser-(O-tert-butyl)-Gly-Phe-Leu-Thr-(O-tert-butyl-OH) (BUBU), deltorphin II, [d-Pen2,d-Pen5]-Enkephalin (DPDPE), the systemically active DPDPE derivative JOM-13, and H-Dmt-Tic-NH-CH2-Bid all produced DOR-mediated antidepressant-like effects in rodent models used to study antidepressant drugs (19, 20). The antidepressant-like effects observed with DPDPE, deltorphin II, H-Dmt-Tic-NH-CH2, and JOM-13 were blocked by the selective DOR antagonist naltrindole, demonstrating these behaviors were mediated through the DOR (20).
Likewise, activating DORs by elevating endogenous opioid levels also produces antidepressant-like effects in animal models. Administration of enkephalinase inhibitors elevate levels of endogenous opioids that act at multiple opioid receptors, but some studies suggest that activity at the DOR mediates this antidepressant action. RB38A (a mixed inhibitor of enkephalinase), RB38B (a selective inhibitor of endopeptidase EC 3.4.24.11), and the orally active enkephalinase inhibitor BL-2401 produced antidepressant-like effects in rodents that were blocked by the non-selective opioid receptor antagonist naloxone, demonstrating an opioid-mediated effect (18, 21, 22). However, several other studies (19, 23–25) demonstrated that the selective DOR antagonist naltrindole could block the antidepressant-like effects produced by the enkephalinase inhibitor RB101 in mice and rats in the learned helplessness model of depression and in the forced swim test. Although administration of muopioid–selective doses of naltrexone and administration of the kappaopioid antagonist nor-BNI did not attenuate the antidepressant actions of RB101, the antinociceptive effects of RB101 were demonstrated to be mediated through the muopioid receptor (25, 26). Overall, these studies strongly suggested that DOR activation specifically produces antidepressant actions.
The nonpeptidic delta-opioid agonists produce antidepressant-like effects in multiple behavioral paradigms in rodents. Broom et al. demonstrated that piperazinyl benzamide compounds such as [(+)-4-[(αR)- α-[(2S,5R)-2,5-dimethyl-4-(2-propenyl)-1-piperazinyl]-(3-methoxyphenyl)methyl]-N,N-diethylbenzamide (SNC80) produce antidepressant-like effects in the forced swim test in mice and rats (27). These findings were corroborated by other studies using the forced swim test in mice and rats (24, 28, 29) and in the olfactory bulbectomized mouse and rat models of depression (30, 31) — all behavioral assays used to evaluate antidepressants. In most cases, the selective DOR antagonist naltrindole attenuated these antidepressant-like effects, confirming a DOR-mediated effect. Currently, the evaluation of nonpeptidic delta-opioid agonists for antidepressant activity has been limited to piperazinyl benzamide compounds. The possible antidepressant activities of structurally diverse delta-opioid agonists either have not been evaluated or have not been reported in the literature to date but would provide useful information as to the generality of the behavioral effect.
Complications With Convulsive Effects
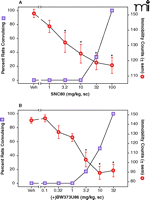
The promising nature and development of nonpeptidic delta-opioid agonists as antidepressants is hindered by the existence of convulsant properties associated with this class of piperazinyl benzamide delta-opioid agonists (Figure 1⇓). In rodents, nonpeptidic agonists produce dose-dependent increases in the number of animals convulsing; these convulsions are brief (lasting < 30 sec) and relatively mild, and are blocked by the delta-opioid antagonist naltrindole (27, 32, 33). In monkeys, the convulsions are less frequent and less predictable, but longer lasting when they do occur; however, no detailed study has fully investigated this effect. Originally, it was proposed that the convulsions might contribute to or generate anti-depressant activity in a manner similar to that observed with electro-convulsive shock in animal studies and in humans. However, several studies and lines of evidence suggest that convulsive properties are not required for the antidepressant-like effects. For example, Broom et al. first demonstrated that the benzodiazepine midazolam blocks the convulsive effects of a delta-opioid agonist without affecting the antidepressant-like effects (34). Midazolam not only blocked the overt convulsive effects, but also prevented all epileptiform activity (35). In addition, tolerance developed rapidly to the convulsive and epileptogenic effects of SNC80 and (+)BW373U86, but not to the antidepressant-like actions, further suggesting that these behavioral outcomes are not necessarily interdependent (34–36).
The effects of the delta-opioid agonists on convulsions and antidepressant-like effects in rats. SNC80 (A) and (+)BW373U86 (B) dose dependently increased convulsions and decreased immobility in the forced swim test, indicating an antidepressant-like effect in rats. For (+)BW373U86, convulsions were observed at the same doses as significant decreases in immobility. With SNC80, significant decreases in immobility were observed prior to the appearance of convulsions. Significant decreases in immobility were determined by one-way ANOVA and Dunnett’s post hoc test. Data were adapted from (34).
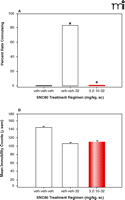
By using an escalating dose regimen, it was demonstrated that the convulsive effects associated with SNC80 administration only could be eliminated completely without altering the antidepressant-like effects of delta-opioid agonist SNC80 (37). Once daily administration of increasing doses of SNC80 completely prevented the convulsive effects of the high dose of SNC80 (Figure 2A⇓); however, this dosing schedule did not alter the level of immobility in the forced swim test as compared with an acute dose (Figure 2B⇓). Convulsions were also decreased dramatically by altering the pharmacokinetic properties with slow intravenous infusions of SNC80; however, the antidepressant-like effects were not changed (38). Overall, these findings demonstrate that the convulsions and antidepressant actions are mediated through the DOR, but the potential therapeutic properties are not contingent on convulsions or non-convulsive seizures. Nonetheless, the following questions still remain: Can DOR-induced antidepressant-like effects exist without the possibility of seizure? Does DOR activation produce seizures or lower seizure thresholds? Is it possible to create a safe nonpeptidic delta agonist with a large therapeutic index?
The effects of increasing doses of the delta-opioid agonist SNC80 on convulsions and antidepressant-like effects in the forced swim test. A single dose of 32 mg/kg SNC80 produced convulsions in 83% of rats (A); however, following once daily administration of increasing SNC80 doses, convulsions were absent, but the decrease in immobility in the forced swim test (i.e., the antidepressant-like effects) remained unchanged. Statistical analysis was described in Figure 1 legend. Data were adapted from (37). veh, vehicle; sc, subcutaneous.
Other classes of nonpeptidic delta-opioid agonists have been evaluated in in vitro and antinociception assays, but there is little information on specific observations for convulsions, pre-convulsive activity, or ictal (i.e., post-seizure) activity. Although overt behavioral convulsions are rarely reported with peptidic delta-opioid agonists [for exception, see (20)], various analogs of endogenous enkephalins and other opioid peptides with some selectivity for the DOR have been shown to produce epileptiform activity and seizures (39–43). Indirect activation of the DOR with the enkephalinase inhibitor RB101 produced significant antidepressant-like effects without producing convulsions or electroencephalographic activity consistent with seizures (25); however, another enkephalinase inhibitor, SCH 32615, demonstrated convulsions in mice at 100-times its minimal effective dose (44). These data might suggest that DOR activation in general may stimulate seizure activity and that complete elimination of convulsions and seizures may be impossible; however, this topic warrants further investigation and better understanding.
It is interesting to consider the actions of delta-opioid agonists in relation to other antidepressant treatments. For example, electro-convulsive therapy is one of the most effective treatments for depression, especially in treatment-resistant and elderly depressed populations. This form of therapy purposely attempts to attain a critical convulsive threshold [for review, see (45)]. Likewise, the antidepressant bupropion (Wellbutrin®) and tricyclic antidepressants have been reported to produce seizures in some patients, and this effect is exaggerated in patients with predispositions for seizure activity (46–48). In fact, one reason for developing the sustained-release formulation of bupropion (Bupropion SR®) was to minimize the risk of seizures [reportedly 5-fold less likely than with the immediate release formulation (49)]. Clinically, seizures need to be minimized for patient safety; however, there appears to be a complex relation between antidepressant action and seizure activity that perhaps should be better understood.
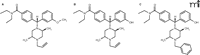
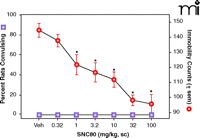
In order to develop a delta-opioid agonist for clinical use, studies would have to demonstrate that there is a large therapeutic window separating the antidepressant-like effects from the convulsive properties and other effects (Box 1). Although ways of minimizing convulsions have been suggested (see above), a compound devoid of convulsant properties would be preferred. Interestingly, a compound based on the piperazinyl benzamide structure (Figure 3⇓), DPI287, produced antidepressant-like effects in the rat forced swim test without convulsions when administered subcutaneously (50). This compound provided a significant improvement in the therapeutic window as compared with structurally similar ligands, with at least a 30- to 100-fold dose separation between the convulsive and antidepressant effects (Figure 4⇓). The isolation of the antidepressant-like properties was only apparent following subcutaneous administration; intravenous administration of DPI-287 produced similar convulsant and antidepressant-like effects as the prototypical agonist SNC80. These data suggested that pharmacokinetic variables play an important role in the convulsive effects of high affinity, high efficacy nonpeptidic delta-opioid agonists.
The structures of some members of the piperazinyl benzamide family. The structures of SNC80 (A), (+)BW373U86 (B), and DPI287 (C) are shown.
The effects of DPI287 on convulsions and antidepressant-like effects in rats. DPI287 significantly decreased immobility in the forced swim test without producing convulsions at any dose tested. Statistical analysis was described in the legend to Figure 1. Data were adapted from (50).
Locomotor Stimulation and Delta-Opioid Agonists
In addition to convulsant properties, delta-opioid agonists also stimulate locomotor activity similar to psychomotor stimulants, such as amphetamine. In many rodent assays used to evaluate antidepressant compounds, stimulant activity identifies a compound as a false-positive; this implies that delta-opioid agonists may not have antidepressant-like effects, but simply general stimulatory effects. Other data suggests, however, that the stimulant activity of delta-opioid agonists is not responsible for the anti-depressant effects. For example, the antidepressant-like effects of SNC80 are still evident after the locomotor-stimulating properties have dissipated (27). Similarly, tolerance develops more rapidly to the stimulant properties of SNC80 as compared with its antidepressant-like effects (37).
Although some stimulant properties are associated with delta-opioid receptor (DOR) activation, one question to consider is: should the presence of stimulant activity in conjunction with antidepressant-like effects prevent the development of delta-opioid agonists as new antidepressant treatments? Bupropion is an antidepressant that mainly increases dopamine signaling by blocking reuptake into the presynaptic terminal and also stimulates locomotor activity in many animal models (65, 66). Similarly, the vigilance-promoting drug modafinil, used for the treatment of narcolepsy, is suggested to have antidepressant actions alone and in combination with typical antidepressants for improved therapeutic results (67). Stimulants in combination with antidepressant drugs or antidepressants with stimulant-like actions (i.e., bupropion, modafinil) are thought to be more effective and faster-acting than antidepressants alone in enhancing recovery and treating residual symptoms, such as fatigue (68–70). This concept is especially encouraging for elderly depressed patients. It is interesting to note that few controlled studies have evaluated the antidepressant properties of stimulants as monotherapy or adjunctive therapy.
Mechanisms Of DOR-mediated Antidepressant Effects
The mechanism responsible for delta-opioid agonist induced anti-depressant-like effects remains unknown. Following DOR activation, what occurs downstream of the receptor to produce these antidepressant actions? Neurotransmitter [i.e., dopamine (DA), serotonin (5HT), or norepinephrine (NE)] signaling may be increased, producing a functionally similar response to known antidepressants; however, the specific identities of the neurotransmitters involved are not known. Using the forced swim test, Detke et al. suggested that changes in certain behaviors indicate increases in specific neurotransmitter systems (51). Treatment with delta-opioid agonists generally increases swimming and climbing behaviors, implying a possible increase in DA, NE, and 5HT. Additionally, delta-opioid agonists produce increases in locomotor activity, conditioned place preference, and antiparkinsonian effects in animal models, suggesting that these compounds increase dopaminergic signaling (33, 52–54); however, the delta-opioid agonists SNC80 and BW373U86 were not self-administered in monkeys and did not stimulate dopamine release as measured by in vivo microdialysis in rats (55, 56). Based on these studies, delta-opioid agonists might indirectly amplify post-synaptic dopamine transmission. Clearly, the interactions between the dopamine and DOR systems are complex and poorly understood, but these interactions may be responsible for the antidepressant action of delta-opioid agonists.
The neurotrophic factor hypothesis is another possible mechanism of action for delta-opioid agonist-induced antidepressant-like effects. Neurotrophins such as brain derived neurotrophic factor (BDNF) may play a role in the efficacy of antidepressants by promoting the growth, maintenance, and plasticity of neurons [for reviews, see (57, 58)]. It was proposed that the therapeutic action of antidepressants requires four to six weeks because neuronal changes occur following antidepressant treatment. Torregrossa and colleagues demonstrated that acute treatment with delta-opioid agonists rapidly increases the expression of BDNF mRNA as compared with mRNA levels observed after acute administration of typical antidepressants (20, 36, 59). Additionally, Narita et al. showed that the delta-opioid agonist SNC80 promoted neural differentiation from neural stem cells obtained from embryonic C3H mouse forebrain (60). These data suggest that delta-opioid agonists might share a common pathway with typical antidepressants, but work on a faster time scale.
More specifically, delta-opioid agonists have neuroprotective effects on dopamine transporters, dopamine neurons, and cortical neurons (61–63); however, reversing the neurotransmitter depletion and possible neurodegeneration associated with depression and chronic stress still needs to be evaluated. Likewise, the delta-opioid agonist SNC80 decreased astrogliosis in the cingulate cortex in mice; accumulated astrocytes in the cingulate cortex are correlated with the generation of negative emotive states especially in aged individuals (64). These data further suggest that DOR activation may produce neurobiological changes associated with enhanced mood or emotional states.
Conclusions
DOR agonists produce a unique pharmacological profile that results in activities differing from those of mu- and kappa-opioid agonists. DOR activation produces antidepressant-like effects in a number of preclinical assays, suggesting that this may be another pharmacological target for treating depression in humans. The development of delta-opioid agonists as antidepressants is hindered by the convulsive activity observed with these compounds, even though it is not responsible for the antidepressant action. Although there are ways to minimize or even eliminate convulsions, a nonpeptidic delta-opioid agonist without any convulsive properties would be ideal. To date, only nonpeptidic delta-opioid agonists from the piperazinyl benzamide class have been reported in the literature for antidepressant-like effects and convulsions; therefore, studies with other classes of delta-opioid agonists would be informative. In general, activation of the DOR system has an interesting behavioral profile distinct from other opioids.
Acknowledgments
The author thanks Dr. James H. Woods at the University of Michigan for supporting and encouraging much of the work presented and cited in this article. This work was supported by grants from the Unites States Public Health Service Grants DA00254, T32 GM07767, and T32 DA07267.
- © American Society for Pharmacology and Experimental Theraputics 2006
References

Emily M. Jutkiewicz, PhD, is a postdoctoral fellow at the University of Michigan in the laboratory of Dr. Margaret E. Gnegy. She received her PhD from the department of Pharmacology at the University of Michigan in 2004 where she trained in the laboratory of Dr. James H Woods. Her PhD thesis work focused on the behavioral effects of delta-opioid agonists in rats. In general, her research interests include the behavioral effects of opioids and other drugs of abuse as well as the pharmacological properties of and the neurobiological changes produced by these compounds. E-mail: ejutkiew{at}umich.edu; fax (734) 764-7118.