 |
 |

Orlistat in the Long-term Treatment of Obesity in Primary Care Settings
Jonathan Hauptman, MD;
Charles Lucas, MD;
Mark N. Boldrin, MS;
Harry Collins, MD
Karen R. Segal, PhD
Arch Fam Med. 2000;9:160-167.
ABSTRACT
 |  |
Objective To evaluate the long-term efficacy and tolerability within primary care settings of orlistat, a gastrointestinal lipase inhibitor, for the treatment of obesity.
Design Randomized, double-blind, placebo-controlled, multicenter study.
Participants A group of 796 obese patients (body mass index, 30-44 kg/m2), treated with placebo 3 times a day (TID), 60 mg of orlistat TID, or 120 mg of orlistat TID, in conjunction with a reduced-energy diet for the first year and a weight-maintenance diet during the second year.
Setting Seventeen primary care centers in the United States.
Main Outcome Measures Changes in body weight and obesity-related disease risk factors.
Results Patients treated with orlistat lost significantly more weight (7.08 ± 0.54 and 7.94 ± 0.57 kg for the 60-mg and 120-mg orlistat groups, respectively) than those treated with placebo (4.14 ± 0.56 kg) in year 1 (P<.001) and sustained more of this weight loss during year 2 (P<.001). More patients treated with orlistat lost 5% or more of their initial weight in year 1 (48.8% and 50.5% of patients in the 60-mg and 120-mg groups, respectively) compared with placebo (30.7%; P<.001), and approximately 34% of patients in the orlistat groups sustained weight loss of 5% or greater over 2 years compared with 24% in the placebo group (P<.001). Orlistat produced greater improvements than placebo in serum lipid levels and blood pressure and was well tolerated, although treatment resulted in a higher incidence of gastrointestinal events.
Conclusions This long-term study indicates that orlistat is an effective adjunct to dietary intervention in the treatment of obesity in primary care settings.
INTRODUCTION
OBESITY IS a chronic medical disorder affecting roughly one third of the population of the United States and is associated with increased morbidity and mortality.1-5 Considerable evidence suggests that weight loss of 5% to 10% of initial body weight substantially improves the health of obese patients.6-9 Diet and exercise, the cornerstones of obesity treatment, have limited effectiveness with respect to long-term weight loss and maintenance.10-12 Several studies suggest that adjunctive pharmacotherapy in combination with medical nutritional therapy improves long-term efficacy; however, some of these medications, now withdrawn, have been associated with serious adverse effects. Nonetheless, there is evidence that one of the most significant benefits of adjunctive pharmacotherapy for obesity lies in the long-term maintenance of weight loss.
Obesity is one of the most common chronic conditions seen by primary care physicians; these physicians treat most obesity in the United States. However, poor long-term success in weight management, time constraints, and the absence of safe and effective drug therapy13-14 prevent primary care physicians from being even more involved in managing obesity. In addition, no studies to date have evaluated pharmacological treatment in primary care settings of individuals who are obese. The present study was undertaken to fill this gap in current knowledge.
Orlistat is a selective and potent inhibitor of gastrointestinal (GI) lipases involved in triglyceride hydrolysis, which restricts the absorption of triglycerides from the GI tract. It has a low systemic absorption.15-16 A drug that alters the absorption of ingested nutrients can potentially induce negative energy balance, assuming incomplete dietary compensation. Orlistat reduces dietary fat absorption in a dose-dependent fashion, with a maximal inhibition of 30% fat absorption at a dosage of 120 mg 3 times a day (TID).15 Short-term, placebo-controlled studies have demonstrated that orlistat produces greater weight loss than diet alone and is well tolerated in obese patients.17-18 The efficacy of orlistat has been further established in a 2-year placebo-controlled trial that combined orlistat with lifestyle intervention by physicans who specialize in treating obesity, and in a 1-year placebo-controlled study of patients with type 2 diabetes treated with oral sulfonylurea medications.19-20 The present study evaluated the efficacy of orlistat in producing and maintaining long-term weight loss, and in improving obesity-related risk factors in individuals who are obese and who received limited dietary and behavioral counseling in a primary care setting.
MATERIALS AND METHODS
STUDY POPULATION
A group of 796 women and men who were obese (age >18 years) with a body mass index (BMI) of 30 to 44 kg/m2 were enrolled. Women were excluded if they were pregnant, lactating, or of childbearing potential and not taking adequate contraceptive measures. Other exclusions were weight loss of more than 4 kg during the previous 3 months; a history of significant cardiac, renal, hepatic, or gastrointestinal disorders; uncontrolled hypertension or any other clinically significant condition; gastrointestinal surgery for weight-reducing purposes; bulimia or laxative and/or substance abuse; abnormal laboratory measures (values 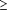 10% greater than the reference value for the normal range and sufficient to require medical follow-up by the study physician); changes in smoking habits in the previous 6 months; or use of any drug that might influence body weight or food intake during the 8 weeks before screening. There was no overlap between the subjects who participated in this trial and subjects in any other clinical trial of orlistat. All patients provided written informed consent. The protocol was approved by the institutional review board at each center and conducted in accordance with the revised Declaration of Helsinki. 10% greater than the reference value for the normal range and sufficient to require medical follow-up by the study physician); changes in smoking habits in the previous 6 months; or use of any drug that might influence body weight or food intake during the 8 weeks before screening. There was no overlap between the subjects who participated in this trial and subjects in any other clinical trial of orlistat. All patients provided written informed consent. The protocol was approved by the institutional review board at each center and conducted in accordance with the revised Declaration of Helsinki.
STUDY DESIGN
This was a randomized, double-blind, parallel-group, placebo-controlled study conducted in 17 primary care centers in the United States. After a 4-week single-blind, placebo lead-in period, 635 patients were randomized to receive placebo, 60 mg of orlistat TID, or 120 mg of orlistat TID with main meals for 52 weeks. All patients were started on a reduced-energy diet at the onset of this 4-week placebo lead-in period and followed this diet during the subsequent 52 weeks of double-blind treatment. Patients then received the same treatment for a second year in combination with a weight-maintenance diet intended to prevent weight regain rather than induce further weight loss. To enter into the double-blind phase, patients must have completed the placebo run-in period with at least 75% compliance, determined by counting the capsules returned by the patients at specified visits and calculating the number of capsules that had been consumed each day.
DIET AND PHYSICAL ACTIVITY
The reduced-energy diet prescribed during the 4-week lead-in period and the first 52 weeks of treatment was nutritionally balanced; supplied about 30% of energy as fat, 50% as carbohydrate, and 20% as protein; and contained a maximum of 300 mg/d of cholesterol. Alcohol consumption was limited to a maximum of 10 drinks per week. The prescribed energy intake was 5020 kJ/d for patients who weighed less than 90 kg initially and 6275 kJ/d for those who weighed 90 kg or more initially. Patients received dietary guidance on the desired energy intake from the study physician only at the start of the placebo lead-in phase. Participating physicians did not receive any specific training in nutrition or weight management techniques beyond the same instructional materials given to the patients (see below).
To evaluate the efficacy of orlistat in prevention of weight regain in year 2, the prescribed energy intake was increased by 1255 kJ/d for patients who were still losing weight at the end of the first 52 weeks of treatment. No dietary adjustment was made for those whose weight remained stable. Registered dieticians or behavioral psychologists were not involved in nutritional or behavioral counseling at any stage of the study. At 4 points during the first 52 weeks, patients viewed videos of behavior modification techniques for weight control. At 4 points during the second year, they were given weight management and diet pamphlets designed to assist in weight maintenance. The written materials were part of the Live for Life program (Johnson & Johnson Health Management Inc, New Brunswick, NJ). No group meetings or counseling sessions were held.
Patients completed 3-day dietary records at 10 points during the study. At week 4, patients received brief instructions on how to complete the food intake records from the physicians and their staff and viewed an instructional video on food records for reinforcement. Throughout the study, patients were encouraged to increase physical activity by walking briskly for 20 to 30 minutes 3 to 5 times per week but were not required to keep records of their physical activity.
ASSESSMENTS
The initial screening assessment consisted of a medical history and physical examination, an electrocardiogram, and measurement of laboratory parameters and body weight. Baseline measurements were made on the day before double-blind treatment began and included body weight; waist circumference; fasting serum glucose, insulin, and lipid levels; routine laboratory parameters; vital signs; adverse events; and an electrocardiogram. A similar assessment, including physical examination, was repeated at the end of 52 and 104 weeks. Interim brief physician visits were scheduled at weeks 1, 2, 8, 16, 24, 32, 44, 60, 76, and 92. Food intake records were analyzed by Professional Nutrition Systems Inc (Westwood, Kan). Results of the food record analyses were not given to the physicians or subjects until they had completed the study.
EFFICACY MEASUREMENTS
The primary efficacy parameter was the change in body weight. Body weight was measured on a calibrated balance scale every 2 weeks for the first month, every 4 weeks until week 52, and every 8 weeks in the second year of the study. The patients wore indoor street clothes and no shoes for the body weight measurement. Patients were weighed at least twice at each assessment until consecutive measurements were within 0.5 kg of each other; the average of the 2 measurements was recorded. Waist circumference was measured with a spring-loaded measuring tape at 24, 52, and 104 weeks, and fasting blood samples were drawn for laboratory determinations after 4, 12, 24, 36, 44, 52, 60, 76, 92, and 104 weeks. Secondary efficacy parameters included serum lipid levels (total cholesterol, low-density lipoprotein [LDL] cholesterol, high-density lipoprotein [HDL] cholesterol, the LDL/HDL ratio, and triglycerides); fasting serum glucose and insulin levels; blood pressure; and waist circumference.
SAFETY AND TOLERABILITY MEASURES
All adverse events were recorded, regardless of their relationship to treatment. To ensure consistency in identifying GI events related to orlistat treatment, a dictionary of standard terms was developed to describe defecation patterns. Laboratory evaluations consisted of hematology; clinical chemistry, including measurements of vitamins A (retinol), D (25-hydroxyvitamin D), and E ( -tocopherol), prothrombin time, and -tocopherol), prothrombin time, and 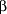 -carotene; urinalysis; a 24-hour urine test; and detection of blood in stools. -carotene; urinalysis; a 24-hour urine test; and detection of blood in stools.
STATISTICAL ANALYSES
Efficacy data were analyzed on an intent-to-treat (ITT) basis (ie, the ITT population was defined as all patients who were randomized to treatment groups, received at least 1 dose of double-blind medication, and had at least 1 follow-up body weight measurement). The null hypothesis for the primary and secondary efficacy parameters was that the expected mean changes after 52 and 104 weeks of double-blind treatment in the patients who received 60 mg or 120 mg of orlistat were not different from those of the patients who received placebo. The significance of treatment differences in the primary efficacy parameters was tested using analysis of variance models that took into account centers, strata, and randomization groups, or, in the case of empty strata, an analysis of covariance model.21 The 95% confidence interval of treatment difference based on the least squares means was also determined. In short, the least squares mean is the expected value of the treatment means for a balanced design with all the covariates at their mean value. This provides a more precise estimate than the arithmetic mean: it takes into account differences that existed between treatments at baseline for the covariates and adjusts for them.22 The last observation carried forward technique was employed for 1- and 2-year analyses, as recommended in the Consolidated Standard of Reporting Trials (CONSORT) statement.23 Briefly, this approach uses all follow-up data, including those obtained from subjects who withdrew prematurely, with the last recorded data point used in the statistical analysis.24 However, all reported data are the actual observed values rather than derived data from carrying forward the last recorded values. The  2 statistic was used to test the significance of differences in proportions. Additional analyses are described in the "Results" section. For all analyses, the P<.05 level of significance was used. Values presented are mean ± SEM unless otherwise indicated. 2 statistic was used to test the significance of differences in proportions. Additional analyses are described in the "Results" section. For all analyses, the P<.05 level of significance was used. Values presented are mean ± SEM unless otherwise indicated.
RESULTS
DISPOSITION OF STUDY PATIENTS AND DEMOGRAPHIC CHARACTERISTICS
Of the 796 patients initially enrolled in the study (Figure 1), 635 (80%) completed the placebo lead-in period. These patients were then randomized to the placebo (n = 212), 60-mg orlistat (n = 213), or 120-mg orlistat (n = 210) group. The demographic characteristics of these 635 patients are shown in Table 1. There were no significant differences in the initial characteristics of the placebo and orlistat treatment groups. Four hundred twenty-seven patients completed the first year of double-blind treatment (122 [57%] received placebo, 154 [72%] received 60 mg of orlistat, and 151 [72%] received 120 mg of orlistat). The percentage of patients who completed 1 year was greater in both orlistat groups than the placebo group (P = .001). All patients who completed the first year were enrolled and had at least one measurement of body weight in the second year. Three hundred twenty-eight patients completed 2 years of double-blind treatment (91 [43%] received placebo, 120 [56%] received 60 mg of orlistat, and 117 [56%] received 120 mg of orlistat). The percentage of patients who completed 2 years of double-blind treatment was also greater in both orlistat treatment groups compared with the placebo group. The most frequent reasons for premature withdrawal during the 2 years of the study (Table 2) were administrative reasons (including change in job, relocation, and family reasons) in the placebo group (17.5%) and loss to follow-up in the orlistat groups (13.1% in the 60-mg group and 15.2% in the 120-mg group).
|
|

|
|
Figure 1. Study design and disposition of subjects.
|
|
|
|
|

|
|
Table 1. Demographic Characteristics (n = 635)*
|
|
|
|
|

|
|
Table 2. Reasons for Premature Withdrawal During 104-Week Double-blind Treatment Period*
|
|
|
BODY WEIGHT CHANGES
Table 3 shows body weight changes during the placebo lead-in period and during the 2 years of double-blind treatment for both the ITT population and the subjects who completed the 2 years of the study. The results for the ITT population and the subjects who completed the 2 full years of the study were similar: across all treatments, the mean weight loss was slightly greater for the subjects who completed the study compared with the ITT subjects; however, the pattern of differences among treatments was identical, regardless of which analysis population was used. Weight loss during the 4-week placebo run-in period was similar in all treatment groups; thereafter, the placebo-treated group lost less weight compared with both orlistat-treated groups (Table 3). Both the 60-mg and 120-mg dosages of orlistat produced significantly greater weight loss than placebo at all points over the 2 years of treatment (P<.001). The largest decrease in weight was observed with the 120-mg dosage of orlistat. At the end of 1 year, the percentage of weight loss from initial body weight was 7.9% ± 0.79% (7.94 ± 0.57 kg) in the 120-mg orlistat group compared with 4.2% ± 0.6% (4.14 ± 0.56 kg) in the placebo group (P<.001). In the second year, the energy intake was increased to maintain body weight rather than induce further weight loss. Maintenance of weight loss was also significantly better with both dosages of orlistat during the second year of treatment, resulting in significant differences from placebo at the end point of 104 weeks (Table 3) (P<.001).
|
|

|
|
Table 3. Initial Body Weight and Changes in Body Weight After 4-Week Placebo Lead-in Period (Day 1) and Over 2 Years of Double-blind Treatment With Orlistat or Placebo*
|
|
|
Analysis of the frequency distribution of weight loss (Figure 2) for the ITT population revealed that 30.7% of patients in the placebo group lost 5% or more of their initial body weight after 1 year, compared with 50.5% in the 120-mg orlistat group (P<.001 vs placebo) and 48.8% in the 60-mg orlistat group (P<.001 vs placebo). The actual weight loss for those patients who lost 5% or more of their initial body weight after 1 year ranged from 11.9 ± 0.65 kg for the 120-mg orlistat group to 9.5 ± 0.51 kg for the placebo group. Weight loss of 10% or more after 1 year was achieved by 11.3% of patients in the placebo group compared with 28.6% of patients in the 120-mg orlistat group (P<.001 vs placebo) and 24.4% in the 60-mg orlistat group (P<.001 vs placebo). A smaller percentage of patients in the placebo group maintained a weight loss of 5% or more of initial body weight at the end of 2 years (24.1%) compared with 34.3% in the 120-mg orlistat group (P = .02 vs placebo) and 33.8% in the 60-mg orlistat group (P = .03 vs placebo). The actual weight loss for patients who lost 5% or more of initial body weight after 2 years ranged from 11.7 ± 0.56 kg for the 120-mg orlistat group to 8.7 ± 0.54 kg for the placebo group. At the end of 2 years, a weight loss of 10% or more of initial body weight was maintained in only 6.6% of patients in the placebo group compared with 18.6% in the 120-mg orlistat group (P = .001 vs placebo) and 14.6% in the 60-mg orlistat group (P = .008 vs placebo). A similar pattern of results was observed for the categorical analysis of the completers and the ITT population, but the absolute percentage of patients achieving 5% or more and 10% or more loss of initial weight was somewhat greater in subjects who completed 2 full years because of the exclusion in the completers analysis of data from dropouts, who may have withdrawn because of lack of weight-loss efficacy. For example, at the end of year 2, 16.5% and 22.8% of subjects who completed 2 years of treatment in the 60-mg and 120-mg orlistat groups, respectively, lost more than 10% of their initial weight compared with 8.8% in the placebo group (P = .10 for the 60-mg orlistat group vs placebo and P = .007 for the 120-mg orlistat group vs placebo).
|
|

|
|
Figure 2. Frequency distribution of weight loss of 5% or greater (top) and 10% or greater (bottom) from initial body weight after 1 year and 2 years of double-blind treatment.
|
|
|
Although patients in all 3 groups regained some weight during the second year of the study, weight regain in year 2 was greater in the placebo group than in both orlistat groups. Percentage of body weight loss at the end of 2 years of treatment was 1.70% ± 0.62% (1.65 ± 0.62 kg) of initial weight in the placebo group, compared with losses of 4.44% ± 0.61% (4.46 ± 0.61 kg) (P<.001) and 5.01% ± 0.79% (5.02 ± 0.73 kg) (P<.001) maintained in the 60-mg and 120-mg orlistat groups, respectively. This result reflects a significantly smaller relapse from weight loss in the patients treated with orlistat. From 52 to 104 weeks, when a weight maintenance rather than a reduced-energy diet was prescribed, the weight regain expressed as a percentage of the weight lost in the first 52 weeks of the study was 60% in the placebo group compared with 37% and 38% in the 60-mg and 120-mg orlistat groups, respectively.
CARDIOVASCULAR RISK FACTORS
Reductions in all lipid parameters were observed in all treatment groups during the placebo lead-in period, which reflects the short-term effect of energy restriction alone (Table 4). After 1 year of double-blind treatment, total cholesterol and LDL cholesterol levels (Table 4) were significantly lower in patients treated with 60 mg and 120 mg of orlistat compared with placebo (P = .001). The effects of orlistat treatment on serum lipids seen during the first year were generally maintained during the second year of treatment. Low density lipoprotein cholesterol levels at 104 weeks (Table 4) were significantly lower in both orlistat groups compared with placebo (P = .03 for placebo vs 60 mg of orlistat and P = .01 for placebo vs 120 mg of orlistat).
|
|

|
|
Table 4. Serum Lipid, Glucose, and Insulin Levels and Blood Pressure Measurements in Study Participants*
|
|
|
Diastolic blood pressure (Table 4) increased slightly in the placebo group (1.1 ± 0.04 mm Hg) during the first year of treatment and decreased in both the 120-mg orlistat group (-0.94 ± 0.02 mm Hg; P = .12) and the 60-mg orlistat group (-0.97 ± 0.01 mm Hg; P = .02). During the second year of treatment, systolic blood pressure was significantly reduced (P = .04) in the 120-mg orlistat group compared with placebo (Table 4), whereas changes in diastolic blood pressure did not differ significantly among groups. There were no significant differences among treatment groups in serum triglyceride or glucose levels at any time. Fasting insulin levels in the 120-mg orlistat group were significantly lower than in the placebo group only after 52 weeks of treatment. A similar pattern of results was obtained by the ITT analysis (Table 4) and the analysis of subjects who completed the study (data not shown) with respect to lipid, glucose, and insulin levels.
TOLERABILITY
In general, adverse events in all groups were transient, mild, or moderate in intensity and resolved without intervention. With the exception of GI events, the incidence and type of adverse events were similar in all treatment groups. Reasons for premature withdrawal from the study are presented in Table 2. Adverse events accounted for the withdrawal of 7.1% (n = 15), 6.6% (n = 14), and 11.0% (n = 23) of subjects in the placebo and 60-mg and 120-mg orlistat groups, respectively, over 104 weeks of double-blind treatment; this was not significantly different among groups. As expected from the mechanism, the incidence of GI events was higher in the orlistat groups. Over 2 years, 59%, 72%, and 79% of patients in the placebo, 60-mg orlistat, and 120-mg orlistat groups, respectively, reported GI events (P = .003 for placebo vs 60 mg of orlistat and P = .001 for placebo vs 120 mg of orlistat). Specific GI events possibly or probably related to orlistat treatment are presented in Table 5, and all of these occurred significantly more frequently in both orlistat groups compared with placebo (P = .001). Most GI events were mild to moderate in intensity, were limited to only 1 or 2 episodes per patient, and occurred early during treatment. Indeed, few GI events were reported during the second year of the study. Ten patients (4.7%) in the 60-mg orlistat group and 12 (5.7%) in the 120-mg orlistat group withdrew from the study prematurely because of GI events, compared with 3 patients (1.4%) in the placebo group.
|
|

|
|
Table 5. Patients With Gastrointestinal Events Potentially Related to Treatment During 2 Years of Treatment, With Incidence Greater Than 5% and Incidence in Any Orlistat Group That Is at Least Twice That of Placebo*
|
|
|
Mean plasma levels of vitamins A, D, and E and 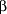 -carotene remained within the reference range in all treatment groups throughout the 2-year study. Vitamin A and vitamin E levels actually increased during the study in all treatment groups, while vitamin D and -carotene remained within the reference range in all treatment groups throughout the 2-year study. Vitamin A and vitamin E levels actually increased during the study in all treatment groups, while vitamin D and 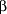 -carotene levels decreased in all treatment groups. Two consecutive low vitamin E and -carotene levels decreased in all treatment groups. Two consecutive low vitamin E and 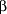 -carotene values occurred more frequently in patients treated with orlistat compared with those given placebo, while the frequency of 2 consecutive low vitamin A and D values was not significantly different among treatment groups (Table 6). Supplementation with -carotene values occurred more frequently in patients treated with orlistat compared with those given placebo, while the frequency of 2 consecutive low vitamin A and D values was not significantly different among treatment groups (Table 6). Supplementation with 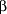 -carotene was required and received by 2.4%, 4.3%, and 6.3% of those in the placebo, 60-mg orlistat, and 120-mg orlistat groups, respectively. Overall, fewer than 1.9% of all the patients required and received vitamin A or E supplementation. Almost all patients who needed vitamin supplementation achieved normal levels by the end of the study. -carotene was required and received by 2.4%, 4.3%, and 6.3% of those in the placebo, 60-mg orlistat, and 120-mg orlistat groups, respectively. Overall, fewer than 1.9% of all the patients required and received vitamin A or E supplementation. Almost all patients who needed vitamin supplementation achieved normal levels by the end of the study.
|
|

|
|
Table 6. Percentage of Patients With 2 or More Consecutive Low Vitamin Levels During 2 Years of Treatment*
|
|
|
COMMENT
Obesity is a major public health problem and is mostly treated by primary care physicians. It is a chronic disorder, like hypertension; consequently, weight loss must be achieved and maintained for a prolonged period to meet both physician and patient goals. However, weight loss is rarely sustained over time when the current weight management approaches are used; indeed, the consensus statement from the National Institutes of Health reported that nearly two thirds of weight loss is regained in the year after initial weight loss.13
Dietary intervention is the most common treatment modality for obesity in the primary care setting. However, while diet and lifestyle intervention alone is effective in producing short-term weight loss (6 months), these treatments rarely result in long-term (1 or 2 years) sustained weight loss or weight maintenance.14 While long-term weight management may be improved by adjunctive antiobesity medication, weight regain also occurs with antiobesity drugs.25-26 For example, in a long-term study of the combination of phentermine and the recently withdrawn fenfluramine, patients who received drug treatment for 156 weeks regained 30% of their lost weight, while those treated with placebo regained 60%, despite continued behavior and diet modification.26 Therefore, there is clearly a need for effective and safe therapeutic options that are accessible to primary care physicians with limited ancillary resources to effect dietary and lifestyle changes.
The present study is the first long-term obesity drug treatment trial conducted in a primary care setting. The results demonstrate that the use of orlistat in this setting produced significantly greater weight loss than placebo after 1 year of treatment. Moreover, weight regain was diminished by orlistat during the second year of treatment, maintaining a significant difference from placebo after 2 years. Greater efficacy was observed with the 120-mg dosage, both with respect to the mean percentage change in body weight and the frequency distribution of weight loss. While this difference may seem small in the present trial, the 120-mg dosage was significantly more effective in other trials, which were statistically powered to test for the difference between dosages. Patient-physician interactions in this study reflect the realistic day-to-day situation that will commonly occur in primary care practice: registered dieticians were not involved in patient counseling, and physicians and their staff provided all information and materials intended to reinforce dietary and lifestyle messages.
In a commentary regarding a 2-year randomized clinical trial of orlistat plus dietary modification,27 Williamson28 noted that the efficacy of weight-loss drugs in the absence of concomitant lifestyle intervention is unknown, but is of potential interest because in actual clinical settings weight-loss medications are likely to be prescribed without an accompanying lifestyle program. The present study approximates these conditions; that is, use of an antiobesity medication without intensive dietary and behavioral counseling while still maintaining an acceptable standard of care. However, one aspect of the study protocol does not approximate actual clinical practice: if patients started to regain weight in year 2, they were instructed not to resume a reduced-energy diet but rather avoid further weight gain. Under realistic clinical practice conditions, patients who relapsed would most likely be encouraged to reduce their energy intake for a period to reverse the weight regain.
Total weight loss over 1 year in the present study was approximately 1 kg less than in a previous investigation,27 which was primarily conducted in academic research centers by obesity specialists. However, the difference between treatment with placebo and orlistat (ie, the treatment effect) was greater in the present study. These observations support the hypothesis that partial inhibition of dietary fat absorption produces a significant energy deficit, leading to weight loss, and this indicates that orlistat is a useful addition to standard dietary therapy in the primary care setting. The weight loss sustained over 2 years in the patients treated with orlistat was sufficient to produce changes in total and LDL cholesterol levels, and the LDL/HDL ratio. These lipid variables improved significantly more in patients treated with 120-mg orlistat compared with placebo after the first year of the study. A progressive rise in these lipid levels during the second year of treatment with placebo was attenuated by orlistat. Decreases in fasting plasma insulin levels and diastolic blood pressure were also greater in the orlistat-treated groups than in the placebo group, although the magnitude of these effects was small and inconsistent. After 2 years of treatment, LDL cholesterol levels remained significantly lower in both orlistat-treated groups compared with placebo, and this likely reflects an independent lipid-lowering effect of orlistat rather than simply a weight-loss effect per se.
Despite substantial evidence for the health benefits of loss of 5% to 10% of initial weight,29 this degree of weight loss fails to meet the treatment expectations of both patient and physician.30 While this point emphasizes the importance of setting realistic goals in obesity treatment, it is also worthy of mention that weight loss of this magnitude may be associated with small changes in several individual obesity-related risk factors; these concomitant changes could substantially improve the overall risk status of cardiovascular disease. Further studies are needed to demonstrate the long-term health consequences and cost benefit of multiple risk factor reduction.
Orlistat was well tolerated. The adverse event profile, characterized by a higher incidence of GI events, was predictable from orlistat's mode of action. However, these events were generally mild to moderate in intensity, usually occurred no more than twice in any patient, and rarely led to the withdrawal of treatment. Orlistat may reduce the efficiency of fat-soluble vitamin absorption from the GI tract.31-32 Consistent with previous studies,27, 31-32 mean levels of vitamins A, D, and E and 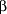 -carotene remained within the normal range in all 3 groups throughout the study in spite of the discontinuation of all vitamin supplements 8 weeks prior to study entry. -carotene remained within the normal range in all 3 groups throughout the study in spite of the discontinuation of all vitamin supplements 8 weeks prior to study entry.
CONCLUSIONS
This large 2-year double-blind placebo-controlled study, the first conducted in the primary care setting, emphasizes the advantage of diet plus pharmacotherapy with orlistat over dietary treatment alone. Although the results may seem to be modest, this study demonstrates that medication can help control weight and lipids over 2 years without extensive nutrition education, exercise training, and behavioral treatment. The addition of more intensive dietary counseling, psychosocial support, and lifestyle modification may produce greater efficacy in some patients. Nevertheless, 120 mg of orlistat provides a well-tolerated means of achieving sustained modest weight loss and improvement in selected risk factors for individuals whose obesity puts their health at risk29 and offers primary care physicians a useful tool to treat obesity.
AUTHOR INFORMATION
Accepted for publication June 28, 1999.
The other members of the US Orlistat Primary Care Study Group are Mark Apte, MD (Future Health Care, Mogadore, Ohio); Robert Bedinger, Jr, MD (McGuire Clinic, Richmond, Va); Kenneth Boren, MD (private practice, Mesa, Ariz); Richard Cannon, MD (Salt Lake City Research Foundation, Salt Lake City, Utah); David Ginsberg, DO (Harleysville Medical Association Inc, Harleysville, Pa); Robert Goldstein, MD (private practice, Tampa, Fla); David Jennings, MD (Research Memphis, Memphis, Tenn); Maureen McCabe, MD (Menninger Clinic, Topeka, Kan); Dennis McClusky, MD (Future Health Care, Mogadore, Ohio); John Orchard, MD (Mercy Hospital of Pittsburgh, Pittsburgh, Pa); Gary Post, MD (Denver Drug Research Inc, Englewood, Colo); Harry Serfer, DO (Family Practice and General Medicine, Hollywood, Fla); Jim Tarro, MD (private practice, Tualatin, Ore).
Reprints: Jonathan Hauptman, MD, Hoffmann–LaRoche Inc, 340 Kingsland St, Nutley, NJ 07110 (e-mail: jonathan.hauptman{at}roche.com).
for the Orlistat Primary Care Study Group
From Departments of International Clinical Research (Dr Hauptman and Mr Boldrin) and Medical Affairs (Drs Lucas and Segal), Hoffman–LaRoche Inc, Nutley, NJ; and the Edison Medical Group, Edison, NJ (Dr Collins).
REFERENCES
 |  |
1. Flegal KM, Carroll MD, Kuczmarski RJ, Johnson CL. Overweight and obesity in the United States: prevalence and trends, 1960-1994. Int J Obes Relat Metab Disord. 1998;22:38-47.
2. Manson JE, Willett WC, Stampfer MJ, et al. Body weight and mortality among women. N Engl J Med. 1995;333:677-685.
FREE FULL TEXT
3. McGinnis JM, Foege WH. Actual causes of death in the United States. JAMA. 1993;270:2207-2212.
FREE FULL TEXT
4. Eckel RH, Krauss RM for the AHA Nutrition Committee. American Heart Association call to action: obesity as a major risk factor for coronary heart disease. Circulation. 1998;97:2099-2100.
FREE FULL TEXT
5. Higgins M, Kannel W, Garrison R, Pinsky J, Strokes III J. Hazards of obesity: the Framingham experience. Acta Med Scand Suppl. 1988;723:23-36.
PUBMED
6. Dattilo AM, Kris-Etherton PM. Effects of weight reduction on blood lipids and lipoproteins: a meta-analysis. Am J Clin Nutr. 1992;56:320-328.
FREE FULL TEXT
7. Goldstein DI. Beneficial health effects of modest weight loss. Int J Obes. 1992;16:397-415.
ISI
| PUBMED
8. Kanders BS, Blackburn GL. Health consequences of therapeutic weight loss: reducing primary risk factors. In: Wadden TA, van Itallie TB, eds. The Treatment of the Seriously Obese Patient. New York, NY: Guilford Press; 1992:213-230.
9. Williamson DF, Pamuk E, Thus M, Flanders D, Byers T, Heath C. Prospective study of intentional weight loss and mortality in never-smoking US white women aged 40-64 years. Am J Epidemiol. 1995;141:1128-1141.
FREE FULL TEXT
10. Safer DJ. Diet, behavior modification and exercise: a review of obesity treatments from a long-term perspective. South Med J. 1991;84:1470-1474.
ISI
| PUBMED
11. Technology Assessment Conference Statement. Methods for voluntary weight loss and control. Ann Intern Med. 1993;119:764-770.
FREE FULL TEXT
12. Wing R, Venditti E, Jakicic J, Polley B, Lang W. Lifestyle intervention in overweight individuals with a family history of diabetes. Diabetes Care. 1998;21:350-360.
ABSTRACT
13. Manson JE, Faich GA. Pharmacotherapy for obesity: do the benefits outweigh the risks? N Engl J Med. 1996;335:659-660.
FREE FULL TEXT
14. Hyman FN, Sempos E, Saltsman J, Glinsmann WH. Evidence for success of caloric restriction in weight loss and control: summary of data from industry. Ann Intern Med. 1993;119:681-687.
FREE FULL TEXT
15. Zhi J, Melia AT, Guerciolini R, Chung J, Kingberg J, Hauptman JR. Retrospective population-based analysis of the dose-response (fecal fat excretion) relationship of orlistat in normal and obese volunteers. Clin Pharmacol Ther. 1994;56:82-85.
ISI
| PUBMED
16. Hauptman JB, Jeunet FS, Hartmann D. Initial studies in humans with the novel gastrointestinal lipase inhibitor Ro 18-0647 (tetrahydrolipstatin). Am J Clin Nutr. 1992;55(suppl 1):309S-313S.
17. Drent ML, Larsson I, William-Olsson T, et al. Orlistat (Ro 18-0647), a lipase inhibitor, in the treatment of human obesity: a multiple dose study. Int J Obes Relat Metab Disord. 1995;19:221-226.
ISI
| PUBMED
18. Drent ML, van der Veen EA. First clinical studies with orlistat: a short review. Obes Res. 1995;3(suppl 4):623S-625S.
19. Sjöström L, Rissanen A, Andersen T for the European Multicentre Orlistat Study Group. Randomised placebo-controlled trial of orlistat for weight loss and prevention of weight regain in obese patients. Lancet. 1998;352:167-172.
FULL TEXT
|
ISI
| PUBMED
20. Hollander PA, Elbein SC, Hirsch IB, et al. Role of orlistat in the treatment of obese patients with type 2 diabetes: a 1-year randomized double-blind study. Diabetes Care. 1998;21:1288-1294.
ABSTRACT
21. Winer BJ. Statistical Principles in Experimental Design. 2nd ed. New York, NY: McGraw-Hill Book Co; 1971.
22. Little R, Yau L. Intent-to-treat analysis for longitudinal studies with drop-outs. Biometrics. 1996;52:1324-1333.
FULL TEXT
|
ISI
| PUBMED
23. Begg C, Cho M, Eastwood S, et al. Improving the quality of reporting of randomized controlled trials: the CONSORT statement. JAMA. 1996;276:637-639.
FREE FULL TEXT
24. Allison DB, Cappelleri JC, Carpenter KM. Design and analysis of obesity treatment and prevention trials. In: Dalton S, ed. Overweight and Weight Management. Gaithersburg, Md: Aspen Publishers Inc; 1997:557-597.
25. Guy-Grand B, Apfelbaum M, Crepaldi G, Gries A, Lefebvre P, Turner P. International trial of long-term dexfenfluramine in obesity. Lancet. 1989;2:1142-1145.
PUBMED
26. Weintraub M, Sundaresan PR, Schuster B, et al. Long-term weight control study, IV (weeks 156-190): the second double-blind phase. Clin Pharmacol Ther. 1992;51:608-614.
ISI
| PUBMED
27. Davidson MH, Hauptman J, DiGirolamo M, et al. Weight control and risk factor reduction in obese subjects treated for 2 years with orlistat: a randomized controlled trial. JAMA. 1999;281:235-242.
FREE FULL TEXT
28. Williamson DF. Pharmacotherapy for obesity. JAMA. 1999;281:278-280.
FREE FULL TEXT
29. National Heart, Lung, and Blood Institute. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. Bethesda, Md: National Institutes of Health; 1998.
30. Foster GD, Wadden TA, Vogt RA, Brewer G. What is a reasonable weight loss? patients' expectations and evaluations of obesity treatment outcomes. J Consult Clin Psychol. 1997;65:79-85.
FULL TEXT
|
ISI
| PUBMED
31. Melia AT, Koss-Twardy SG, Zhi J. The effect of orlistat, an inhibitor of dietary fat absorption, on the absorption of vitamins A and E in healthy volunteers. J Clin Pharmacol. 1996;36:647-653.
ABSTRACT
32. Zhi J, Melia AT, Koss-Twardy SG, Arora S, Patel IH. The effect of orlistat, an inhibitor of dietary fat absorption, on the pharmacokinetics of beta-carotene in healthy volunteers. J Clin Pharmacol. 1996;36:152-159.
ABSTRACT
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(2):175-177.
FULL TEXT
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
 |
Over-the-counter weight loss with orlistat?
DTB 2009;47:125-127.
ABSTRACT
| FULL TEXT
Differences between human subjects in the composition of the faecal bacterial community and faecal metabolism of linoleic acid
Devillard et al.
Microbiology 2009;155:513-520.
ABSTRACT
| FULL TEXT
Mechanism of conjugated linoleic acid and vaccenic acid formation in human faecal suspensions and pure cultures of intestinal bacteria
McIntosh et al.
Microbiology 2009;155:285-294.
ABSTRACT
| FULL TEXT
REVIEW PAPER: Current Strategies in the Development of Anti-obesity Drugs and Their Safety Concerns
Elangbam
Vet Pathol 2009;46:10-24.
ABSTRACT
| FULL TEXT
Review: Pharmacotherapy as part of a weight management programme: a UK perspective
Barnett
British Journal of Diabetes & Vascular Disease 2007;7:268-277.
ABSTRACT
State of the Art Review: Long-term Pharmacotherapy for Overweight and Obesity: A Review of Sibutramine, Orlistat, and Rimonabant
Dunican et al.
AMERICAN JOURNAL OF LIFESTYLE MEDICINE 2007;1:367-388.
ABSTRACT
Pharmacotherapeutic Options for Overweight Adolescents
Dunican et al.
The Annals of Pharmacotherapy 2007;41:1445-1455.
ABSTRACT
| FULL TEXT
Rimonabant: A novel selective cannabinoid-1 receptor antagonist for treatment of obesity
Patel and Pathak
Am J Health Syst Pharm 2007;64:481-489.
ABSTRACT
| FULL TEXT
Pharmacologic Treatment Options for Obesity: Current and Potential Medications
Fujioka and Lee
Nutr Clin Pract 2007;22:50-54.
ABSTRACT
| FULL TEXT
Low-Dose Orlistat Effects on Body Weight of Mildly to Moderately Overweight Individuals: A 16 Week, Double-Blind, Placebo-Controlled Trial
Anderson et al.
The Annals of Pharmacotherapy 2006;40:1717-1723.
ABSTRACT
| FULL TEXT
Cannabinoid-1 Receptor Antagonist, Rimonabant, for Management of Obesity and Related Risks
Gadde and Allison
Circulation 2006;114:974-984.
FULL TEXT
Obesity in older adults: technical review and position statement of the American Society for Nutrition and NAASO, The Obesity Society
Villareal et al.
Am. J. Clin. Nutr. 2005;82:923-934.
ABSTRACT
| FULL TEXT
A Comparison of Meal Replacements and Medication in Weight Maintenance after Weight Loss
LeCheminant et al.
J. Am. Coll. Nutr. 2005;24:347-353.
ABSTRACT
| FULL TEXT
Use And Costs Of Bariatric Surgery And Prescription Weight-Loss Medications
Encinosa et al.
Health Aff (Millwood) 2005;24:1039-1046.
ABSTRACT
| FULL TEXT
Effects of Weight Loss in Overweight/Obese Individuals and Long-Term Hypertension Outcomes: A Systematic Review
Aucott et al.
Hypertension 2005;45:1035-1041.
ABSTRACT
| FULL TEXT
Meta-Analysis: Pharmacologic Treatment of Obesity
Li et al.
ANN INTERN MED 2005;142:532-546.
ABSTRACT
| FULL TEXT
Changes in body weight and serum lipid profile in obese patients treated with orlistat in addition to a hypocaloric diet: a systematic review of randomized clinical trials
Hutton and Fergusson
Am. J. Clin. Nutr. 2004;80:1461-1468.
ABSTRACT
| FULL TEXT
Clinical Implications of Obesity With Specific Focus on Cardiovascular Disease: A Statement for Professionals From the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: Endorsed by the American College of Cardiology Foundation
Klein et al.
Circulation 2004;110:2952-2967.
ABSTRACT
| FULL TEXT
Diagnosis and Treatment of Obesity in Adults: An Applied Evidence-Based Review
Orzano and Scott
J Am Board Fam Med 2004;17:359-369.
ABSTRACT
| FULL TEXT
Screening and Interventions for Obesity in Adults: Summary of the Evidence for the U.S. Preventive Services Task Force
McTigue et al.
ANN INTERN MED 2003;139:933-949.
ABSTRACT
| FULL TEXT
Anesthetic Considerations for Bariatric Surgery
Ogunnaike et al.
Anesth. Analg. 2002;95:1793-1805.
FULL TEXT
Effects of Obesity Surgery on Non-Insulin-Dependent Diabetes Mellitus
Greenway et al.
Arch Surg 2002;137:1109-1117.
ABSTRACT
| FULL TEXT
Obesity
Yanovski and Yanovski
NEJM 2002;346:591-602.
FULL TEXT
Evidence-Based Nutrition Principles and Recommendations for the Treatment and Prevention of Diabetes and Related Complications
Franz et al.
Diabetes Care 2002;25:148-198.
FULL TEXT
Extracts from ""Clinical Evidence"": Obesity
Arterburn and Noel
BMJ 2001;322:1406-1409.
FULL TEXT
Targeted Advertising in Medical Journals
Clemenson and Bowman
Arch Fam Med 2000;9:791-791.
FULL TEXT
Effects of Weight Loss With Orlistat on Glucose Tolerance and Progression to Type 2 Diabetes in Obese Adults
Heymsfield et al.
Arch Intern Med 2000;160:1321-1326.
ABSTRACT
| FULL TEXT
|