 |
 |

Spontaneous Splenic Infarction Secondary to Diabetes-Induced Microvascular Disease
Peter P. Tóth, MD, PhD;
Robert K. Reuter, DO;
James MacDonald, DO
Arch Fam Med. 2000;9:195-197.
ABSTRACT
Splenic infarction is a clinical entity seldom encountered. The most frequent causes of splenic infarction include thromboembolic phenomena, hematologic malignant neoplasms, and vasculitides. We describe a patient who sustained splenic infarction secondary to diabetes-induced, small-vessel atherosclerotic disease.
INTRODUCTION
Splenic infarction is a clinical entity that has received increasing clinical attention in recent years. Although it is seldom encountered, splenic infarction has many potential causes. The most frequent causes of splenic infarction include myelofibrosis and hematologic malignant neoplasms.1 Other causative factors include thromboembolic disease stemming from atrial fibrillation2 and thrombus situated along the thoracic aorta,3 rheumatologic disorders including Kawasaki disease4 and systemic lupus erythematosus complicated by the presence of lupus anticoagulant and anticardiolipin antibodies,5 falciparum malaria,6 adult respiratory distress syndrome,7 rupture of splenic artery aneurysm,8 septic emboli in endocarditis,9 sickle cell disease,10 and Wegener granulomatosis.11 In these various conditions, splenic ischemia is induced either suddenly with massive occlusion or vascular discontinuity, or gradually as infiltration of the splenic parenchyma with hematopoietic elements occurs and its blood supply is strangulated.
Splenic infarction can be either symptomatic or asymptomatic. In one series of 59 cases, 41 patients (69%) had symptoms on presentation, including fever, chills, left upper quadrant pain, and constitutional symptoms, eg, malaise, nausea, and early satiety.1 When splenic infarction occurs secondary to septic emboli in the setting of endocarditis, up to 90% of patients have no localizing findings.9 The radiographic characteristics of splenic infarction with either ultrasonography12 or computed tomography (CT)11, 13 are well defined, though CT has emerged as the preferred imaging modality for making the diagnosis.
Among diabetic patients, there are 2 case reports of patients developing splenic infarction secondary to embolic phenomena while receiving peritoneal dialysis for end-stage renal disease.14 The objective of this article is to report a case of splenic infarction secondary to intrasplenic microvascular disease in an insulin-dependent diabetic patient. Based on a MEDLINE search of the medical literature since 1966 using the terms "diabetes mellitus" and "splenic infarction" in MeSH format and as text words, to our knowledge, this is the first such case reported in the medical literature to date.
REPORT OF A CASE
A 37-year-old white woman was seen in the emergency department with a 1-week history of steadily crescendoing left-sided flank and left upper quadrant discomfort. On the day of presentation the pain began to extend into the left lower quadrant as well. She had been experiencing fevers, chills, and nausea with one episode of vomiting during the previous day. She denied dysuria, urgency, or frequency. She had a bowel movement on the day of admission to the hospital. Admission vital signs were as follows: blood pressure, 129/76 mm Hg; pulse, 102 beats/min; respirations 16/min; and temperature, 36.6°C.
The patient's medical history was significant for juvenile-onset diabetes mellitus at the age of 4 years. Diabetes-related complications included bilateral blindness since 1984 secondary to hemorrhage and severe proliferative diabetic retinopathy, diabetic neuropathy in the hands and feet, and diabetic gastroparesis. She was hypothyroid but receiving adequate oral levothryroxine sodium (Synthroid) replacement therapy (0.3 mg/d). Psychiatric history was significant for depression, anxiety, trichotillomania, and premenstrual dysphoric disorder. Her pharmacologic regimen included subcutaneous extended insulin zinc (Ultralente), 20 U every morning; human insulin lispro (Humalog) according to a sliding scale in the evening; oral sertraline hydrochloride (Zoloft), 100 mg/d; oral clonazepam (Klonopin), 0.5 mg twice daily; oral lisinopril (Zestril), 5 mg/d; and cisapride (Propulsid), 10 mg, before meals and at bedtime. She smoked 1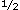 packs of cigarettes per day and had done so for at least 20 years. packs of cigarettes per day and had done so for at least 20 years.
Physical examination revealed left upper quadrant tenderness with more mild left lower quadrant tenderness. There was mild tenderness to percussion of the left flank. Bowel sounds were present. There was neither rebound or guarding nor splenomegaly. Peripheral pulses were diminished. The patient's leukocyte count was 10.8 x 109/L, with neutrophil predominance (0.93) without left shift. The serum hemoglobin level was 1270 g/L; the platelet count was 24.9 x 109/L. Urinalysis showed trace protein with no leukocytes, erythrocytes, or leukocyte esterase. The levels of serum electrolytes, serum urea nitrogen, creatinine, amylase, creatine kinase, and liver function test results were all within normal limits. Glucose level was 8.2 mmol/L (147 mg/dL) with no evidence of diabetic ketoacidosis; the hemoglobin A1c level was 0.07 (7.5%). Serum human chorionic gonadotropin was negative for pregnancy. Stool was negative for occult blood. Abdominal radiographs showed a nonspecific ileus. Ultrasonographic evaluation of the kidneys revealed no evidence of abscess, hydronephrosis, or dilatation of the collecting system. Abdominal contrast-enhanced CT scan disclosed a wedge-shaped region of low attenuation within the central portion of the spleen consistent with splenic infarction (Figure 1). Findings from a peripheral smear of her blood were normal. Transesophageal echocardiography demonstrated normal cardiac anatomy and function; no evidence of thrombus was noted in any of the cardiac chambers or within the ascending or descending portions of the aorta.
|
|

|
|
Figure 1. Computed axial tomogram demonstrating wedge-shaped defect in the central portion of the spleen with apex directed toward the hilum.
|
|
|
The patient's condition was stabilized and she was given intravenous narcotic analgesia and fluid. Her blood glucose levels were controlled with sliding scale insulin therapy. During hospitalization the patient remained afebrile, her leukocyte counts normalized, and she received adequate oral analgesia. The decision was made to monitor her condition further and forestall splenectomy because most patients achieve spontaneous resolution of symptoms with time. Anticoagulation therapy was considered but later rejected because of her blindness from diabetic retinopathy. She was given a pneumococcal vaccine (Pneumovax) and was discharged home in stable and afebrile condition with well-controlled blood glucose levels.
During the next 10 days, the patient required steadily escalating doses of oral meperidine (Demerol) hydrochloride to achieve adequate analgesia. Because her pain had become intractable, it was decided that the patient warranted splenectomy; this was performed by laparotomy. The patient's left upper quadrant and left flank pain resolved subsequent to surgery. Gross examination of the spleen revealed multiple small triangular infarctions with their apices directed toward the hilum. Microscopic examination of these infarcted areas revealed occlusive small-vessel atherosclerotic disease (Figure 2).
|
|

|
|
Figure 2. Light micrograph of an artery near the apex of the splenic infarction. The vessel has marked narrowing of its lumen secondary to small-vessel atherosclerotic disease. (hematoxylin-eosin, original magnification x100).
|
|
|
COMMENT
Given the fact that diabetes mellitus causes diffuse small-vessel atherosclerotic disease, it is somewhat surprising that spontaneous splenic infarction is not more common in patients with diabetes and in patients with peripheral vascular disease. To our knowledge, this is the first such case reported. In 2 fairly large series (a total of 82 cases), no cases of splenic infarction secondary to diabetes-induced, small-vessel atherosclerosis were reported.1, 12 These studies found that the most common causes of splenic infarction include myelofibrosis, hematologic malignancy (including leukemia, lymphoma, or plasmacytomas), thromboembolic disease (stemming from atrial fibrillation, myocardial infarction, or aortic thrombus), and sepsis secondary to endocarditis.
The CT scan was strongly suggestive of splenic infarction. Other diagnostic possibilities include hematoma, abscess, and neoplasm. A wedge-shaped region of low attenuation with the apex directed toward the splenic hilum is essentially diagnostic for splenic infarction.13 In contrast, panarteritis caused by inflammatory diseases such as Wegener granulomatosis and polyarteritis nodosa gives rise to more diffuse infarction and broader, more encompassing areas of low attenuation on CT scanning.11 In our patient the conclusion that her CT scan was consistent with splenic infarction is supported by the fact that she had no recent history of trauma, and she was afebrile. Her leukocyte count normalized within 24 hours of hospital admission, her complete blood cell counts, and peripheral blood smear result were inconsistent with a hematologic malignant neoplasm.
The initial decision to postpone surgery and follow up the patient's condition is clearly justified by the clinical outcomes reported by others.1, 12 Intractable pain, splenic rupture, and abscess formation are clear indications for splenectomy. This patient's spleen contained multiple small infarcts in regions of occlusive microvascular atherosclerotic disease. It is possible that her pain worsened and became intractable because she experienced a series of splenic infarctions over a short period. This case highlights the importance of considering atherosclerotic disease of the native splenic small vasculature in diabetic patients since hematologic malignant neoplasm, thromboembolic disease, and other potential causes of splenic infarction were all ruled out.
AUTHOR INFORMATION
Accepted for publication May 27, 1999.
Corresponding author: Peter P. Tóth, MD, PhD, Sullivan Clinic, Sarah Bush Lincoln Health System, 7 Hawthorne Ln, Sullivan, IL 61938.
From the Sullivan Clinic, Sarah Bush Lincoln Health System, Sullivan, Ill (Dr Tóth); and the Departments of Pathology (Dr Reuter) and Surgery (Dr MacDonald), Sarah Bush Lincoln Health Center, Mattoon, Ill.
REFERENCES
 |  |
1. Nores M, Phillips EH, Morgenstern L, Hiatt JR. The clinical spectrum of splenic infarction. Am Surg. 1998;64:182-188.
ISI
| PUBMED
2. Argiris A. Splenic and renal infarctions complicating atrial fibrillation. Mt Sinai J Med. 1997;64:342-349.
PUBMED
3. Agolini SF, Shah KT, Goodreau JJ, McLoughlin TM, Sinclair MC. Splenic infarction caused by a large thoracic aortic thrombus. J Vasc Surg. 1997;26:1069-1072.
FULL TEXT
|
ISI
| PUBMED
4. Huong D, Gatfosse M, Papo T, Barjonet G, Lacombe P, Godeua P. Symptomatic splenic infarction complicating adult Kawasaki disease. Br J Rheumatol. 1995;34:579-586.
FREE FULL TEXT
5. Arnold MH, Schrieber L. Splenic and renal infarction in systemic lupus erythematosus: association with anti-cardiolipin antibodies. Clin Rheumatol. 1988;7:406-410.
FULL TEXT
|
ISI
| PUBMED
6. Agarwal VK, Agarwal S, Pathak T. Splenic infarct in falciparum malaria. Indian J Pediatr. 1997;34:1050-1051.
7. Lo AY, Reich H, Harvey J. Splenic infarction associated with adult respiratory distress syndrome. Mt Sinai J Med. 1994;61:369-371.
PUBMED
8. Jacobson IV, Crowe PJ. Splenic infarction: a complication of splenic artery aneurysm. Aust N Z J Surg. 1994;64:53-54.
ISI
| PUBMED
9. Ting W, Silverman NA, Arzouman DA, Levitsky S. Splenic septic emboli in endocarditis. Circulation. 1990;82(suppl IV):105-109.
10. Wetton CWN, Tran TL. Case report: splenic infarct in sickle cell disease. Clin Radiol. 1995;50:573-574.
FULL TEXT
|
ISI
| PUBMED
11. Fonner BT, Nemcek AA, Boschman C. CT appearance of splenic infarction in Wegener's granulomatosis. Am J Radiology. 1994;164:353-354.
12. Georg C., Schwerk WB. Splenic infarction: sonographic patterns, diagnosis, follow-up, and complications. Radiology. 1990;174:803-807.
FREE FULL TEXT
13. Merrick RD, Mehta JB, Cowan J, Donahue D, Young M. CT scan of the abdomen in the evaluation of splenic infarction. Tenn Med. 1990;83:77-78.
14. Syed A, Holley JL, Piraino B. Splenic infarct presenting as sterile peritonitis with peripheral embolic phenomena. Adv Perit Dial. 1993;9:202-205.
PUBMED
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(2):175-177.
FULL TEXT
|