 |
 |

Demographic Predictors of Melanoma Stage at Diagnosis
Daniel J. Van Durme, MD;
Jeanne M. Ferrante, MD;
Naazneen Pal, MPH;
Deanna Wathington, MD, MPH;
Richard G. Roetzheim, MD, MSPH;
Eduardo C. Gonzalez, MD
Arch Fam Med. 2000;9:606-611.
ABSTRACT
 |  |
Objective To examine sociodemographic characteristics as possible predictors of late-stage melanoma diagnosis. We hypothesized that late-stage diagnosis would be associated with the following: older age, male sex, unmarried status, lower educational attainment and income level, rural residence, and cigarette smoking.
Methods We used data from the state tumor registry to study all incident cases of melanoma occurring in Florida during 1994 whose stage at diagnosis was available (N=1884). We used multiple logistic regression to determine the effects of sociodemographic characteristics on the odds of late-stage (regional or distant metastases) diagnosis.
Results There were 243 patients (12.9%) diagnosed as having melanoma that had metastasized to either regional lymph nodes or distant sites. Patients who were unmarried (odds ratio, 1.5; P=.01), male (odds ratio, 2.2; P<.001), or smokers (odds ratio, 2.2; P<.001) or who resided in communities with lower median educational attainment (odds ratio, 1.5; P=.048) had greater odds of having a late-stage diagnosis.
Conclusions To detect these cancers at an earlier stage and improve outcomes, there should be increased educational efforts directed toward physicians who treat these patients. A recognition that there may be additional risk factors for late-stage diagnosis, beyond the established risk factors, such as family history and excess sun exposure, should be included in the initial assessment. Specific public education efforts should also be targeted to these patients to increase their self-surveillance and surveillance of their partners.
INTRODUCTION
DURING 1973-1989, the incidence of malignant melanoma increased 85%, more than any other major cancer.1-2 There were approximately 42,000 new cases in 1998, with 7000 deaths.3 The lifetime risk of acquiring melanoma is now estimated to be about 1 in 87.4 Within the next 10 years, the risk is expected to climb to 1 in 75.
The prognosis of malignant melanoma is heavily dependent on the stage at which it is detected.5 Early thin lesions are almost entirely curable.6 However, the 5-year survival for patients with melanomas that have spread to regional lymph nodes is 54% and for those with metastatic disease, only 13%.1 Therefore, understanding the determinants of late-stage melanoma diagnosis is an important cancer-control objective.
Factors predictive of later stage at diagnosis and poor prognosis in previous studies are lower socioeconomic status,7-11 male sex,8-9,12-15 older age,8-10,13-14,16-19 nonwhite race,8, 12, 20 and smoking.21-24
In a previous study,25 we reported the effects of insurance payer and race or ethnicity on cancer stage at diagnosis. For patients diagnosed as having melanoma, we found that Medicaid patients had 4.7 times the odds of having a late-stage diagnosis and that uninsured patients had 2.6 times the odds of having a late-stage diagnosis compared with patients with commercial indemnity insurance. No racial or ethnic differences in stage at diagnosis were identified. In this study, we explored whether other sociodemographic characteristics were predictive of late-stage diagnosis. We hypothesized that a late-stage melanoma diagnosis would be associated with older age, male sex, unmarried status, lower educational attainment and income level, rural residence, and cigarette smoking.
SUBJECTS AND METHODS
SOURCES OF DATA
The study population consisted of all incident cases of melanoma occurring in Florida from January 1, 1994, to January 1, 1995, whose stage at diagnosis was available (N=1884). Incident cases were identified from the Florida Cancer Data System (FCDS), Florida's population-based statewide cancer registry. The FCDS was created in 1978 and has been collecting cancer incidence data since 1981. It is a member of the North American Association of Central Cancer Registries. The FCDS has well-established methods to find all cases, including cooperative arrangements with other state tumor registries and standardized procedures for quality control. The North American Association of Central Cancer Registries audits estimated that the completeness of case ascertainment for all cancers during 1990-1994 was 97% (Lydia Voti, oral communication, January 11, 2000).
The FCDS is primarily a hospital-based reporting system. Hospital sources are discharge diagnostic codes in medical records, hospital pathology reports (inpatient and outpatient), radiation therapy departments, and outpatient departments (day surgery and chemotherapy clinics). Cases are also reported from private laboratories and from practicing physicians; however, finding cases in these settings is probably less complete.
To include information that is not available from the FCDS (insurance payer and socioeconomic status), cases were linked with state inpatient and outpatient discharge abstracts and the 1990 US Census. The state of Florida, Agency for Health Care Administration abstracts discharge data for all admissions to nonfederal acute care hospitals, licensed ambulatory surgical centers, freestanding radiation therapy centers, and diagnostic imaging centers. Abstracted data include patient identifying information (Social Security number, date of birth, and race or ethnicity), discharge diagnoses (up to 10), and insurance payer. The calendar year 1994 is the most recent year for which all the relevant data from both the FCDS and Agency for Health Care Administration are available.
The FCDS cases were matched with the Agency for Health Care Administration inpatient and outpatient discharge abstracts by a probabilistic method using all shared identifying variables (Social Security number, sex, race or ethnicity, and date of birth). Cases that successfully matched on all variables were considered valid matches. Cases were also considered valid matches if the sole discrepancy was a Social Security number or date of birth that differed by only 1 digit. Using this method, 1425 staged cases (75.6%) were successfully matched, a rate comparable to that of similar studies.26 For the remaining 459 cases, we relied solely on information found in the FCDS.
The FCDS cases were next matched with 1990 US Census data to obtain census-derived measures of socioeconomic status. For 95.3% of cases we were able to determine the census tract based on the street address. For the remaining 4.7% of cases, we relied on the patient's ZIP code. Therefore, patients were assigned the median income and education level of either the census tract or ZIP code of their residence. We also used US Census data to determine if the patient's place of residence was urban or rural. Patients were classified as having an urban residence if their ZIP code was designated as entirely urban by the US Census. Patients were classified as having a rural residence if their ZIP code contained at least some portion that was designated as outside urban or rural by the US census. The use of census-derived measures of socioeconomic status has been validated in previous studies.27-29
VARIABLES
The main outcome examined, stage at diagnosis, was defined as the summary stage at the time of diagnosis using the SEER (Surveillance, Epidemiology, and End Results) Program's Summary Staging Guide.30 The summary stage is based on a combination of pathological, operative, and clinical assessments and uses all information available within 2 months of diagnosis. The stage at diagnosis is reported using categories similar to those of the SEER Program: in situ, local, regional (direct extension of disease beyond organ of origin or spread to regional nodes), or distant. For purposes of analysis, the stage at diagnosis was reclassified as either early stage (in situ or local) or late stage (regional or distant).
The stage at diagnosis was available for 1884 (93.6%) of the 2012 incident cases of melanoma occurring in Florida in 1994. Patients who did not undergo staging were older (P=.01) and had lower education (P=.001) and income (P<.001) levels than patients who underwent staging (Wilcoxon rank sum test). There were no differences in race or ethnicity (P=.63), marital status (P=.09), or sex (P=.08) between staged and unstaged cases ( 2 test or Wilcoxon rank sum test). 2 test or Wilcoxon rank sum test).
The following variables were assessed as possible predictors of late-stage melanoma diagnosis: age, sex, marital status (currently married or unmarried), smoking status (current smoker or nonsmoker), education level (high school graduate or less or more than high school), household income (5 levels: <$15,000; $15,000-$24,999; $25,000-$34,999; $35,000-$49,999; and 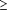 $50,000), and urban vs rural residence. $50,000), and urban vs rural residence.
STATISTICAL ANALYSIS
Relationships between categorical variables and stage at diagnosis were assessed with the  2 test. The comparison of age differences and stage at diagnosis was assessed with the t test. The multivariate relationship between demographic predictors and the odds of late-stage diagnosis was examined using multiple logistic regression. Logistic models included age (as a continuous variable) and indicator variables for marital status, smoking status, education level, income level, and urban vs rural residence. The patient's insurance payer and race or ethnicity were also included in all logistic models so that reported odds ratios were adjusted for these characteristics as well. In addition to their main effects, we also examined interactions between statistically significant variables in subsequent logistic models. Because the malignant potential of some in situ melanomas is uncertain, we also repeated logistic models using only the data from patients with invasive disease. 2 test. The comparison of age differences and stage at diagnosis was assessed with the t test. The multivariate relationship between demographic predictors and the odds of late-stage diagnosis was examined using multiple logistic regression. Logistic models included age (as a continuous variable) and indicator variables for marital status, smoking status, education level, income level, and urban vs rural residence. The patient's insurance payer and race or ethnicity were also included in all logistic models so that reported odds ratios were adjusted for these characteristics as well. In addition to their main effects, we also examined interactions between statistically significant variables in subsequent logistic models. Because the malignant potential of some in situ melanomas is uncertain, we also repeated logistic models using only the data from patients with invasive disease.
Adjusted odds ratios and 95% confidence intervals are reported for each predictor variable. The statistical significance of predictor variables was assessed using the  2 likelihood ratio test.31 All reported P values are 2-tailed. Statistical significance was set at 2 likelihood ratio test.31 All reported P values are 2-tailed. Statistical significance was set at  =.05. =.05.
RESULTS
The patient characteristics are presented in Table 1. The mean (SD) age of the patients was 62.4 (16.6) years, with a range of 13 to 99 years. Most patients with melanoma were male. There were 243 patients (12.9%) diagnosed as having melanoma that had metastasized to either regional lymph nodes or distant sites. Table 2 describes bivariate predictors of late-stage melanoma diagnosis. Late-stage diagnosis was more common among patients who were male, unmarried, and smokers and resided in communities with low median income and education levels. There was a statistically insignificant trend for patients residing in rural communities to be diagnosed at later stage than patients from urban settings. Patients diagnosed as having late-stage disease were similar in age to those diagnosed as having early-stage disease (60.6 vs 62.3 years; t test, 1.51; P=.13).
|
|

|
|
Table 1. Characteristics of 1884 Patients Diagnosed as Having Melanoma in Florida, 1994*
|
|
|
|
|

|
|
Table 2. Predictors of Late-Stage Diagnosis of Melanoma*
|
|
|
Multivariate predictors of late-stage melanoma diagnosis are presented in Table 3. Patients who were unmarried, male, and smokers and resided in communities with a median educational attainment of high school or less were more likely to be diagnosed as having melanoma at a late stage. The stage at diagnosis was unrelated to the patient's age, community measures of median income, or urban vs rural residence. There were no statistically significant interactions between sex, marital status, smoking status, or education level in the logistic regression models. The results of logistic regression analysis were also unchanged when in situ cases were excluded from analysis.
|
|

|
|
Table 3. Multivariate Predictors of Late-Stage Melanoma Diagnosis in 1470 Patients*
|
|
|
COMMENT
We found that the following sociodemographic groups were more likely to be diagnosed as having melanoma at a late stage: male patients, unmarried patients, smokers, and those who resided in communities with low median educational attainment. Among these factors, male sex and smoking had the most pronounced effects, with more than double the odds of late-stage diagnosis.
Our finding that males were more likely to have melanoma diagnosed at a late stage agrees with findings in other studies.8-9,12-15 Men are more likely than women to present with nodular melanomas and melanomas located on the back.14 Tumors in less visible body areas are significantly thicker at the time of diagnosis than those occurring in more highly visible areas, most likely as a result of delayed detection.32-34 Consistent with this view, a study by Koh et al35 found that men were less likely than women to identify their own melanoma. In addition, nodular melanomas are less likely to be detected during visits to physicians for unrelated problems and instead are discovered more often by patients when lesions become symptomatic.32 Men are also less likely to report undergoing either self-screening or physician screening.6, 36 In addition, men are also less knowledgeable about melanoma and have less favorable attitudes.4, 37
The finding that unmarried persons were more likely to be diagnosed as having late-stage melanoma is also consistent with the finding that patients had difficulty detecting less visible lesions on self-examination. Studies by Koh et al35 confirmed the importance of a spouse in detecting many melanomas. In their study, a spouse was the third most common person (after the index patient and a physician) to detect a melanoma. This finding suggests that physician surveillance of melanoma is especially important for unmarried patients.
Similar to the findings in other studies, we found that smokers were more likely to be diagnosed as having melanoma at late stage.21-24 Among the variables studied, smoking was the strongest predictor of a diagnosis of melanoma at a late stage. It has been postulated that smoking leads to a diminished host-immune response against melanomas, leading to greater tumor virulence. It is also possible that smoking is a marker for less active skin surveillance, which would also lead to later detection of melanomas.
We found that patients residing in areas with a low median educational attainment were more likely to have a late-stage diagnosis. Others9-10 similarly found that patients from more affluent areas generally have thinner melanomas at the time of detection. Moreover, patients with melanoma with a lower socioeconomic status are more likely to die of their disease.7-8,11 Patients with a lower socioeconomic status are less likely to undergo either self-screening or physician screening.6, 36 Lower socioeconomic status has also been associated with less knowledge and awareness of melanoma.4 In the results of multivariate analysis, we found an effect for education level, but not for income.
We found no significant linear or nonlinear relationship between age and stage at diagnosis. Most studies9-10,13-14,16-19 have concluded that elderly patients are more likely to be diagnosed at a late stage. The higher frequency of thick melanomas in older patients has been attributed to an increased proportion of nodular melanomas and a decreased ability to recognize the changes of melanoma.14 Our finding that there was no relationship between age and stage at diagnosis may be peculiar to Florida, with its large number of elderly retirees from other states. One would expect that elderly persons who are able to retire to another state are generally healthier, better educated, and better off financially than those who are not able to retire out of state. Miller et al4 found that elderly patients are more likely to do self-examinations.
This study has a number of important limitations. Initially, socioeconomic status was measured at a community not an individual level. However, previous studies27-29,38 have validated the use of aggregate measures of socioeconomic status. We also were limited to the SEER Program's categories of staging (in situ, local, regional, and distant) rather than more detailed staging measures, such as Breslow thickness or Clark level. As cancers are detected and managed increasingly in the outpatient setting, it will become more difficult for tumor registries to find cases. There are no well-established mechanisms for gathering the detailed information in the FCDS about outpatients treated in physicians' offices. Late-stage melanomas are probably more likely than early-stage lesions to be reported to tumor registries.39 An additional concern of skin cancer screening is that greater surveillance could lead to the detection of in situ melanomas with uncertain malignant potential.40-41 Our findings were similar, however, when we analyzed only cases of invasive melanomas. Finally, our study was restricted to incident cases of malignant melanoma in Florida, which may not be representative of other diseases or other parts of the country.
In conclusion, we found that males, unmarried patients, smokers, and those who resided in communities with low median educational attainment were more likely to be diagnosed as having melanoma at a late stage. There are several implications of these findings. Although most physicians know that family history and excess sun exposure are risk factors for melanoma, they should be aware of the other factors that may increase the likelihood of late-stage cancer and should screen patients meeting these criteria more conscientiously. Primary care physicians should screen patients with a higher risk whenever they are in the office, even if the complaints are unrelated to melanoma. More physicians have been trained to recognize risk factors for cardiovascular disease. The same attention should be focused on melanoma detection. Many physicians use problem lists and flow sheets on their office charts. These charts could include an indication as to whether the skin was examined fully and whether self-examination information was given to patients with a higher risk of melanoma.
The American Cancer Society and the American Academy of Dermatology have launched public education efforts to increase awareness of skin cancer. These efforts should be targeted to patients with a higher risk using appropriate messages and the media. Information should recommend increased self-surveillance and checking the partner's skin.
AUTHOR INFORMATION
Accepted for publication October 22, 1999.
Richard G. Roetzheim, MD, MSPH, was supported by a Generalist Physician Faculty Scholars Award from the Robert Wood Johnson Foundation, Princeton, NJ.
Corresponding author and reprints: Daniel J. Van Durme, MD, University of South Florida, Department of Family Medicine, 12901 Bruce B. Downs Blvd, Box 13, Tampa, FL 33612 (e-mail: dvandurm{at}com1.med.usf.edu).
From the Department of Family Medicine (Drs Van Durme, Ferrante, Wathington, Roetzheim, and Gonzalez and Ms Pal) and the Division of Cancer Control, H. Lee Moffitt Cancer Center & Research Institute (Drs Ferrante and Roetzheim), University of South Florida, Tampa.
REFERENCES
 |  |
1. Miller B, Ries L, Hankey B, Kosary C, Edwards B. Cancer Statistics Review: 1973-1989. Bethesda, Md: US Dept of Health and Human Services; 1992. NIH publication 92-278.
2. Friedman RJ, Rigel DS, Silverman MK, Kopf AW, Vossaert KA. Malignant melanoma in the 1990s: the continued importance of early detection and the role of physician examination and self-examination of the skin. CA Cancer J Clin. 1991;41:201-226.
ABSTRACT
3. Landis S, Murray T, Bolden S, Wingo P. Cancer statistics, 1998. CA Cancer J Clin. 1998;48:6-29.
ABSTRACT
4. Miller DR, Geller AC, Wyatt SW, et al. Melanoma awareness and self-examination practices: results of a United States survey. J Am Acad Dermatol. 1996;34:962-970.
FULL TEXT
|
ISI
| PUBMED
5. Ahmed I. Malignant melanoma: prognostic indicators. Mayo Clin Proc. 1997;72:356-361.
ABSTRACT
6. Girgis A, Campbell EM, Redman S, Sanson-Fisher RW. Screening for melanoma: a community survey of prevalence and predictors. Med J Aust. 1991;154:338-343.
ISI
| PUBMED
7. Geller AC, Miller DR, Lew RA, Clapp RW, Wenneker MB, Koh HK. Cutaneous melanoma mortality among the socioeconomically disadvantaged in Massachusetts. Am J Public Health. 1996;86:538-544.
FREE FULL TEXT
8. Chang AE, Karnell LH, Menck HR for the American College of Surgeons Commission on Cancer and the American Cancer Society. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. Cancer. 1998;83:1664-1678.
FULL TEXT
|
ISI
| PUBMED
9. Bonett A, Roder D, Esterman A. Epidemiological features of melanoma in South Australia: implications for cancer control. Med J Aust. 1989;151:502-504, 506-509.
ISI
| PUBMED
10. Roder DM, Luke CG, McCaul KA, Esterman AJ. Trends in prognostic factors of melanoma in South Australia, 1981-1992: implications for health promotion. Med J Aust. 1995;162:25-29.
ISI
| PUBMED
11. Vagero D, Persson G. Risks, survival and trends of malignant melanoma among white and blue collar workers in Sweden. Soc Sci Med. 1984;19:475-478.
12. Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California cancer registry data, 1988-93. Cancer Causes Control. 1997;8:246-252.
FULL TEXT
|
ISI
| PUBMED
13. Hersey P, Sillar RW, Howe CG, et al. Factors related to the presentation of patients with thick primary melanomas. Med J Aust. 1991;154:583-587.
ISI
| PUBMED
14. Hanrahan PF, Hersey P, D'Este CA. Factors involved in presentation of older people with thick melanoma. Med J Aust. 1998;169:410-414.
ISI
| PUBMED
15. Thorn M, Ponten F, Bergstrom R, Sparen P, Adami HO. Clinical and histopathologic predictors of survival in patients with malignant melanoma: a population-based study in Sweden. J Natl Cancer Inst. 1994;86:761-769.
FREE FULL TEXT
16. Austin PF, Cruse CW, Lyman G, Schroer K, Glass F, Reintgen DS. Age as a prognostic factor in the malignant melanoma population. Ann Surg Oncol. 1994;1:487-494.
FULL TEXT
|
ISI
| PUBMED
17. Goodwin JS, Samet JM, Key CR, Humble C, Kutvirt D, Hunt C. Stage at diagnosis of cancer varies with the age of the patient. J Am Geriatr Soc. 1986;34:20-26.
ISI
| PUBMED
18. Hanrahan PF, Hersey P, Watson AB, Callaghan TM. The effect of an educational brochure on knowledge and early detection of melanoma. Aust J Public Health. 1995;19:270-274.
ISI
| PUBMED
19. Holmes F, Hearne E. Cancer stage-to-age relationship: implications for cancer screening in the elderly. J Am Geriatr Soc. 1981;29:55-57.
ISI
| PUBMED
20. Vayer A, Lefor AT. Cutaneous melanoma in African-Americans. South Med J. 1993;86:181-182.
ISI
| PUBMED
21. Koh H, Sober A, Day CJ, Lew R, Fitzpatrick T. Cigarette smoking and malignant melanoma: prognostic implications. Cancer. 1984;53:2570-2573.
FULL TEXT
|
ISI
| PUBMED
22. Rigel D, Friedman R, Levine J, Kopf A, Levenstein M. Cigarette smoking and malignant melanoma: prognostic implications. J Dermatol Surg Oncol. 1981;7:889-891.
ISI
| PUBMED
23. Shaw H, Milton G, McCarthy W, Farago G, Dilworth P. Effect of smoking on the recurrence of malignant melanoma. Med J Aust. 1979;1:208-209.
ISI
| PUBMED
24. Shaw H, Milton G. Smoking and the development of metastases from malignant melanoma. Int J Cancer. 1981;28:153-156.
ISI
| PUBMED
25. Roetzheim R, Pal N, Tennant C, et al. Effects of health insurance and race on the early detection of cancer. J Natl Cancer Inst. 1999;91:1409-1415.
FREE FULL TEXT
26. Ayanian J, Kohler B, Abe T, Epstein A. The relation between health insurance coverage and clinical outcomes among women with breast cancer. N Engl J Med. 1993;329:326-331.
FREE FULL TEXT
27. Hofer T, Wolfe R, Tedeschi P, McMahon L, Griffith J. Use of community versus individual socioeconomic data predicting variation in hospital use. Health Serv Res. 1998;33:243-259.
ISI
| PUBMED
28. Krieger N. Overcoming the absence of socioeconomic data in medical records: validation and application of a census-based methodology. Am J Public Health. 1992;82:703-710.
FREE FULL TEXT
29. Krieger N, Fee E. Social class: the missing link in U.S. health data. Int J Health Serv. 1994;24:25-44.
ISI
| PUBMED
30. Shambaugh E, Weiss M. Summary Staging Guide: Cancer Surveillance Epidemiology and End Results Reporting: SEER Program. Bethesda, Md: National Cancer Institute; 1977.
31. Hosmer D, Lemeshow S. Applied Logistic Regression. New York, NY: John Wiley & Sons; 1989.
32. Temoshok L, DiClemente RJ, Sweet DM, Blois MS, Sagebiel RW. Factors related to patient delay in seeking medical attention for cutaneous malignant melanoma. Cancer. 1984;54:3048-3053.
FULL TEXT
|
ISI
| PUBMED
33. Hemo Y, Gutman M, Klausner JM. Anatomic site of primary melanoma is associated with depth of invasion. Arch Surg. 1999;134:148-150.
FREE FULL TEXT
34. Temoshok L. Biopsychosocial studies on cutaneous malignant melanoma: psychosocial factors associated with prognostic indicators, progression, psychophysiology and tumor-host response. Soc Sci Med. 1985;20:833-840.
35. Koh HK, Miller DR, Geller AC, Clapp RW, Mercer MB, Lew RA. Who discovers melanoma? patterns from a population-based survey. J Am Acad Dermatol. 1992;26:914-919.
ISI
| PUBMED
36. Koh HK, Geller AC, Miller DR, Caruso A, Gage I, Lew RA. Who is being screened for melanoma/skin cancer? characteristics of persons screened in Massachusetts. J Am Acad Dermatol. 1991;24:271-277.
ISI
| PUBMED
37. Brandberg Y, Bolund C, Michelson H, Mansson-Brahme E, Ringborg U, Sjoden PO. Perceived susceptibility to and knowledge of malignant melanoma: screening participants vs the general population. Prev Med. 1996;25:170-177.
FULL TEXT
|
ISI
| PUBMED
38. Diez-Roux A. Bringing context back into epidemiology: variables and fallacies in multilevel analysis. Am J Public Health. 1998;88:216-222.
FREE FULL TEXT
39. Melia J, Frost T, Graham-Brown R, et al. Problems with registration of cutaneous malignant melanoma in England. Br J Cancer. 1995;72:224-228.
ISI
| PUBMED
40. Swerlick RA, Chen S. The melanoma epidemic: is increased surveillance the solution or the problem? Arch Dermatol. 1996;132:881-884.
FREE FULL TEXT
41. Swerlick RA, Chen S. The melanoma epidemic: more apparent than real? Mayo Clin Proc. 1997;72:559-564.
ABSTRACT
RELATED ARTICLE
The Archives of Family Medicine Continuing Medical Education Program
Arch Fam Med. 2000;9(7):639-641.
FULL TEXT
THIS ARTICLE HAS BEEN CITED BY OTHER ARTICLES
The Influence of Marital Status on Stage at Diagnosis and Survival of Older Persons With Melanoma
Reyes Ortiz et al.
Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2007;62:892-898.
ABSTRACT
| FULL TEXT
Distance to Diagnosing Provider as a Measure of Access for Patients With Melanoma
Stitzenberg et al.
Arch Dermatol 2007;143:991-998.
ABSTRACT
| FULL TEXT
|