Systemic Administration of Naked DNA: Gene Transfer to Skeletal Muscle
- 633 Salk Hall, Center for Pharmacogenetics School of Pharmacy University of Pittsburgh Pittsburgh, Pa 15213
- Address correspondence to LH. E-mail Huangl{at}msx.upmc.edu; fax 412-648-1664.
Abstract
Skeletal muscle is a promising target tissue for the gene therapy of both muscle and non-muscle disorders. Gene transfer into muscle tissue can produce a variety of physiologically active proteins and may ultimately be applied to the treatment of many diseases. A variety of methods have been studied to transfer genes into skeletal muscle, including viral and non-viral vectors. In this review, we discuss recent developments in the non-viral delivery of genes to muscles.
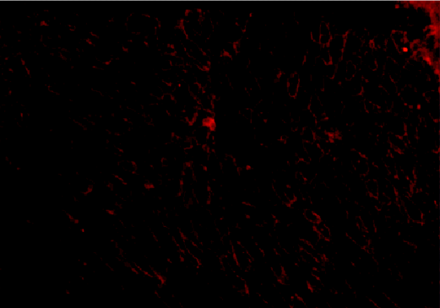
The systemic delivery of naked DNA to within diseased organs offers both advantages and challenges for gene therapy. The diaphragm
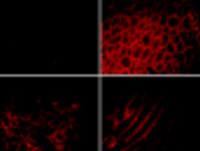
tissue shown above comes from mutant (mdx) mice that are deficient in dystrophin. Subsequent to receiving therapeutic DNA via intravascular injection, however, the
mice express dystrophin (immunostained red) beneath cell membranes.
INTRODUCTION
The use of skeletal muscle as a target organ for gene transfer is a rapidly developing area of gene therapy. In the past ten years, investigations of gene transfer into muscle have progressed from the expression of plasmid reporter genes (1) to the production of therapeutic proteins such as neurotrophic factors (2, 3) and hormones (4–8). For instance, intramuscular administration of an adenovirus vector encoding neurotrophin-3 (AdNT-3) has been tested in the mouse mutant pmn (progressive motor neuronopathy). AdNT-3–treated pmn mice manifest prolonged lifespan, improved neuromuscular function, reduced motor axonal degeneration, and efficient re-innervation of muscle fibers (2).
More recently, an expression plasmid encoding rat erythropoietin (EPO) has been investigated for the correction of anemia associated with renal failure. Electroinjection of the EPO-expressing construct into various rat muscles sustains long-term EPO production and secretion. The muscle-produced EPO alleviates anemia in approximately 80% of nephrectomized rats used to model renal failure (8).
Several factors make skeletal muscle an attractive route for gene transfer: i) The tissue is abundant, making up about 40% of the body weight of an average adult; ii) skeletal muscle is accessible to most of the delivery systems currently used for gene transfer; and iii) there is no significant cell replacement in skeletal muscle tissue, so that introduced genes are not rapidly lost following mitosis, and transgene expression can thus persist for relatively long periods (9, 10).
Gene transfer methods commonly use recombinant viral vectors derived from human adenovirus and adeno-associated virus (AAV) because of the ability of these vectors to transduce large numbers of muscle fibers. However, a few drawbacks to viral vectors have been identified in the past few years. First, the size of genes that can be inserted into the viral genome without disabling gene transfer is limited; for instance, the AAV genome can accommodate only up to 4.8 kb of exogenous sequence (11). Second, despite being replication deficient, there are concerns about the safe use of viruses as therepeutics, including the possibility of recombination with endogenous virus, which could lead to the formation of a deleterious infectious agent. Third, many types of viral vectors, including the commonly used retrovirus, adenovirus, and herpes simplex virus (12), induce an immune response against intrinsic viral antigens, which causes serious toxicity to the host and renders repetitive treatment problematic. Finally, viral vectors are biological agents. Their production requires living cells, and biological systems are difficult to adopt for the good manufacturing practice and quality control procedures mandated by the Food and Drug Administration (13). Although these problems may be overcome eventually, they have prompted efforts in developing alternative (i.e., non-viral) gene delivery systems.
Non-viral systems can be classified into two categories (14): synthetic vectors including cationic lipids and polymers, and naked plasmid DNA. Naked DNA in particular is receiving considerable attention with regard to gene transfer into muscle tissue due to its safety, simplicity, and reliability. Gene transfer by naked plasmid DNA to the skeletal muscle was first demonstrated by intramuscular injection (15). In addition, the large-scale purification of plasmid DNA can be easily accomplished by column chromatography to produce highly purified DNA, the quality of which can be easily controlled. In addition, DNA is much more stable than other delivery vectors and can be refrigerated for long periods of time.
Unfortunately, the transfection efficiency of naked DNA by intramuscular injection is relatively low and establishes transgene expression at the injection site alone, which would not suffice for the treatment of diseases such as hemophilia and muscular dystrophy. Nevertheless, the method has been somewhat successful in regard to genetic vaccination, whereby low-level gene expression is often sufficient to stimulate the immune system (16). In contrast, many muscle diseases, such as muscular dystrophy, would require therapeutic gene expression in most, if not all, muscles in the body. Thus, plasmid DNA would have to be administered systemically in order to achieve effective gene therapy for these diseases. This review addresses the feasibility of gene transfer, through systemic delivery of DNA, into skeletal muscle.
EXPERIMENTAL METHODOLOGIES
Pressure-Mediated Gene Transfer to Limb Musculature
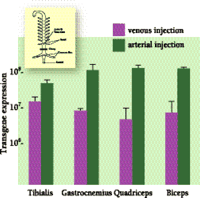
Pressure-mediated gene transfer into muscles was first reported, in 1998, by Budker et al., who found that plasmid DNA can be delivered to and expressed in the hindlimb muscles of rats, with rapid injection of a voluminous DNA solution, under the condition that all blood vessels leading to and out of the hindlimb are occluded (17). Although this method represents a significant advance towards gene transfer to muscle, the technique requires the complicated microsurgery of each leg of the rat. More recently, we have used a simpler surgical procedure for the efficient, semi-systemic transfer of genes to all muscles in both hindlimbs of mice. Specifically, the abdomen is opened prior to injection, and a clamp is placed on the aorta and vena cava just below the kidneys. In such a way, the injected DNA passes through the common iliac arteries and reaches the muscles of both hindlimbs to result in gene expression in all pertinent major muscle groups (Figure 1⇓).
Transgene expression in hindlimb muscles pursuant to intravascular injection of naked DNA. Plasmid DNA (100 mg in 2 ml PBS) that encoded the luciferase gene as a reporter was rapidly (within 5 s) injected through either tail vein or tail artery, and the blood flow in the aorta and vena cava was blocked for 15 s (see inset). Transgene expression in the major muscle groups was determined by luciferase activity 48 h after injection (relative light units). Luciferase activity following intra-arterial injection was about tenfold higher than that following intravenous injection, perhaps reflecting deeper distribution of arteries within muscles.
The endothelium wall represents a barrier for intravascular gene delivery. The endothelium in muscle is continuous and non-fenestrated, showing low permeability to solute and large macromolecules (18). The hydrodynamic pressure induced by fast injection of a large volume of DNA solution, however, effectively overcomes this barrier, probably by expanding the endothelium and thereby making pores accessible for DNA entry. Still, the circumvention of the epithelial barrier cannot ensure that myocytes will take up the DNA that reaches them. Indeed, although fluorescence-labeled plasmid DNA is observed to surround almost all muscle fibers shortly (~5 min) after injection, the majority remains extracellular. This result indicates that cellular uptake of naked DNA will be the major barrier for gene delivery to skeletal muscles despite the pressure-mediated circumvention of the endothelial barrier.
Circulatory Occlusion and Gene Transfer to the Diaphragm
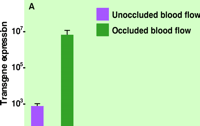
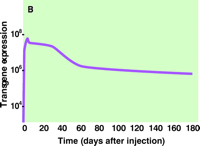
Recently, we have shown that the delivery of reporter and therapeutic genes to the diaphragm is facilitated by transiently blocking blood flow through the diaphragm following intravenous injection of plasmid DNA (19). Specifically, mice were intravenously (tail vein) injected with DNA containing a reporter gene (luciferase), after which blood flow through the diaphragm was immediately occluded by placing a clip on the vena cava below the diaphragm. As shown in Figure 2A⇓, the occlusion of blood flow was necessary to observe significant gene expression. Intriguingly, even brief occlusion (~4 s) is effective, and further prolongation of the time of occlusion does not improve gene expression. Figure 2B⇓ indicates the level of reporter gene activity in the diaphragm to peak at six days, followed by a slow decline of gene expression.
Efficacy of transgene expression in the mouse diaphragm following circulatory occlusion.
A. Circulatory occlusion greatly enhances gene transfer. Blood flow through the diaphragm was blocked (4 s) immediately following intravenous injection of plasmid DNA (80 mg in 100 ml of saline solution) containing a gene for luciferase. The level of gene expression (relative light units per mg of total protein extracted from tissue) was determined one day after injection.
B. Expression of transgene over time. Levels of luciferase activity attained through circulatory occlusion dropped off logarithmically over the two-month period following treatment to reach a value that persisted until at least six months after treatment. The level of gene expression in other organs including the liver, lung, heart, spleen, and kidney was undetectable at day six after administration.
The mechanism of plasmid DNA uptake by the diaphragm muscle through systemic delivery is not clear. Postulated mechanism of plasmid DNA uptake include both receptor-mediated and nonspecific processes. Work in this area has largely been done in isolated hepatocytes (20), and suggests that naked DNA may be taken up in a receptor-mediated process, although the unidentified receptor appears to bind DNA with low affinity. Little information is available for muscle, particularly with systemically delivered DNA, but based on the studies of hepatocytes, we conclude that the successful uptake of DNA arises from the longer exposure time provided by the occlusion of blood flow.
Occlusion of blood flow does not seem to enhance gene transfer to limb muscles; as mentioned above, gene transfer to limb muscles requires pressure-mediated penetration of DNA through the endothelium barrier. It is unclear why gene transfer is achieved by blockage of blood flow to the diaphragm but not to limb muscles. Our data suggest that the endothelium in the diaphragm must not be a significant barrier for DNA, despite functional and biochemical investigations that indicate the it to be similar to that of other skeletal muscles with comparable fiber composition (21, 22). Conceivably, the diaphragm engenders special features that promote blood flow so as to ensure the life-long sustained contraction and relaxation of during each breathing cycle. Such features may include high degrees of muscular vascularization via the microvascular network, and a high content of mitochondria and myoglobin. These characteristics could thus render the diaphragm especially sensitive to circulatory occlusion.
GENE THERAPY OF DUCHENNE MUSCULAR DYSTROPHY
Duchenne muscular dystrophy (DMD) is a recessive genetic disorder (23). DMD is caused by the absence of dystrophin (24), a 427-kd protein encoded on the short arm of the X chromosome by the largest gene currently known (25). The disease causes proximal muscle weakness and may result in the delay of motor development. Affected boys are wheelchair bound at approximately ten years of age and usually die from respiratory failure in their third decade (23).
The delivery of dystrophin-encoding cDNA to the affected muscle tissue is a potential cure for DMD. In attempts to develop this therapeutic strategy, gene transfer into muscle has been evaluated in animal models using direct injection of the cDNA-containing vector into muscle. Most approaches to DMD gene therapy have employed the mdx mouse model, in which a spontaneous mutation of the dystrophin gene has led to the obliteration of dystrophin expression (26). Using pressure-mediated gene transfer, we have delivered a dystrophin gene into leg muscles of mdx mice. Dystrophin-positive fibers become evident in mdx mice seven days after injection of a plasmid containing full-length murine dystrophin cDNA under the control of a CMV promoter. The therapeutic expression of the dystrophin protein is widespread, occurring in all muscle groups of both hind legs.
Because respiratory failure and subsequent death associated with DMD result in part from progressive degeneration of the diaphragm muscle, a potential treatment paradigm would be direct gene transfer into the diaphragm. Compared with hindlimb muscles, the diaphragm is readily accessed neither by direct injection nor by the pressure-mediated approach. More recently, we have elicited significant gene transfer to the diaphragm by circulatory occlusion pursuant to intravenous injection. The restoration of dystrophin expression at the cell membranes of mdx diaphragm muscle fibers, following this protocol, is evident in Figure 3⇓; approximately 40% of all diaphragm muscle fibers become dystrophin-positive. Significantly, the effective transfer of the gene to mdx mice dystrophin shows, upon histological examination, that the expression and assembly of the transgene-encoded dystrophin in the cell membrane of the diaphragm fibers prevents the progression of muscle degeneration and regeneration.
CONCLUSIONS
Gene transfer into skeletal muscle holds promise for the treatment of a variety of serum protein deficiencies, muscular dystrophies, and chronic ischemic limb syndromes. The unique advantage of the systemic delivery of naked DNA is that all muscle fibers within the organism can be potentially accessed so that therapeutic levels of protein can be expressed. Although the current methods remain experimental, the levels of transgene expression achieved to date are promising for certain clinical applications.
Acknowledgments
Work from our labortory was partially supported by NIH grant AR 45925 and by a grant from the Muscular Dystrophy Association of America.
- © American Society for Pharmacology and Experimental Theraputics 2001