Obesity, Body Image and Diet Drugs
100 Years of Change
- Stanley Scheindlin, DSc
“Let me have men about me that are fat.” —Shakespeare, Julius Caesar, Act 1, Scene 2
Caesar would find few to agree with his preference for fat men in twenty-first century America. Obesity has become a subject of great concern among the public and the medical profession, as it is said to have reached epidemic proportions in the United States. As a known risk factor for diabetes, hypertension, coronary artery disease, gall bladder disease, and sleep apnea, obesity is indirectly responsible for a great deal of illness and mortality (1). And even more associations between obesity and adverse conditions are being reported. One recent study finds that an obese woman’s chance of becoming pregnant decreases as her Body Mass Index (BMI) increases (2). Another reports that overweight or obese adults are 50% more likely to have asthma than normal weight adults (3).
“Central obesity,” or excessive fat around the abdomen is associated with the “metabolic syndrome,” a new medical construct. Also known as insulin resistance syndrome, metabolic syndrome is thought to be a precursor of type 2 diabetes, and a risk factor for heart disease and stroke. There are five components of the metabolic syndrome (Box 1); the presence of three or more of them is diagnostic of the condition (4). Of course, not everyone who weighs a few pounds more than that advocated by the standard weight-height table is obese. The key determinant of obesity is the Body Mass Index (BMI), calculated by dividing an individual’s weight (in kilograms) by the square of the height (in meters). A BMI value between 18.5 and 24.9 is considered optimal; overweight is defined as a BMI of 25 to 29.9; and obesity as a BMI of 30 or above (4). BMI is not faultless either: a muscular person will tend to have a higher BMI because muscle weighs more than adipose tissue.
Criteria Comprising the Metabolic Syndrome
-
Abdominal obesity: waist circumference >102 cm (men), >88 cm (women)
-
Hypertriglyceridemia
-
Low HDL cholesterol
-
High blood pressure
-
Fasting glucose 110 mg/dL or higher
Adapted from (4).
Perceptions of Obesity and Body Image
Although fatness is considered undesirable and harmful today, it was not always so. For example, in the 1930s it was arguable whether obesity should be considered a disease or simply a cosmetic issue. If the latter, then weight-reducing products were cosmetics and not subject to FDA regulation. This specific loophole blocked any governmental action against a very toxic product for several years. During the past 100 years, American perceptions of overweightness, obesity, and body image have undergone many changes. Some of these perceptual changes, as well as some of the medications used to achieve weight loss, are discussed in these Reflections.
In the early years of the twentieth century, fatness did not bear a negative connotation. Tuberculosis (TB), or “consumption,” was rampant, and people were painfully familiar with the pallor and emaciation that it caused. A degree of chubbiness was therefore seen as a sign of good health. The ideal of feminine beauty was a full-chested, broad-hipped woman. Women wore corsets, which made the waist seem smaller and the chest fuller. They wore bustles under their skirts, causing the hips to appear larger (5). Among males, there was the stereotype of the jovial fat man, an idea which still survives in our conception of Santa Claus. A moving World War I era story by Rex Beach was titled “Too Fat to Fight.” In this book, the hero tries to enlist in the US Army, but is turned down because of his weight. He travels to France on his own, like a one-man USO, to dispense comforts and cheeriness among the troops in the trenches, and dies a heroic death (6). The jolly, good-natured fat man is now considered to be a myth. The fat person is felt to experience at least as much tension, frustration, and anger as anyone else. Psychologists state that these anxieties drive many people to seek release by nibbling and noshing, thereby putting on excess weight (7).
In women, who, it may be argued, tend to be more concerned with their appearance than men, the ideal body image has seen more changes over the years. In the first two decades of the twentieth century, young women began to rebel against the Victorian era’s ideas of beauty, and against the era’s confining undergarments. The “flappers” of the 1920s favored waistless, sheathlike dresses, concealing their curves and producing a straighter line appearance (5). So desirable was the boyish look, that many women of that era became habitual dieters. The goal of a slimmer body remained in vogue in the Depression years of the 1930s. In the 1940s, during World War II, many women went to work in the war industries, projecting a sturdier, rather than fragile, image. Still, the overall goal of slimness remained.
A new feminine ideal emerged in the late 1940s and 1950s. Exemplified by film star Marilyn Monroe, this ideal featured curves and ample breasts. In the late 1950s, this body image was espoused by a new demographic group. Immigrants from Europe, who had survived the Nazi death camps or otherwise suffered extreme privation during six years of war, were delighted to find abundant food available in the United States, and had no desire to remain thin. Plotz (7), discussing the influence of cultural factors on the management of obesity, noted that these recent immigrants valued plumpness (“zaf-tig”), both for esthetic reasons and as evidence that they had emerged from poverty.
Drugs for Weight Loss
Although, at certain times and in certain circumstances, fatness has been valued, a preoccupation with losing weight has been constant during the past hundred years. There is a large weight loss “industry,” made plainly evident by the many magazine articles and books on dieting, the meal substitutes on the market, and herbal supplements promising effortless weight loss. Only two life style changes are needed for weight loss: reducing food intake and increasing physical activity. This approach is simple, but far from easy. Thus, there has been an ongoing search for pharmacologic help in losing weight. Many drugs have been brought forward for this purpose, each new one being greeted with enthusiasm and each revealing its drawbacks in the fullness of time.
Pageant Winners Reflect Contemporary Body Shape Preferences
Winners of the annual Miss America pageant provide an insight into the ideal of feminine beauty at any period in the twentieth century. Over the decades, American views of what constitutes an ideal female physique have changed significantly. In the Roaring Twenties, Miss Americas often resembled the flappers, whereas during World War II and in the 1950s, most pageant winners possessed heftier shoulders, bosoms, and hips, like many trend-setting movie stars of the time. In the 1960s, despite the popularity of stick-like entertainers such as Twiggy, curvy Miss Americas still prevailed. Miss America of 1969 was athletic and muscular, perhaps reflecting American society’s new interest in physical fitness (15). However, excessive pursuit of athletic prowess, with its emphasis on muscular development and lower weight, can result in what has been dubbed the “female athlete triad”: eating disorder, amenorrhea, and osteoporosis (16). The final three decades of the century again saw an exaggerated worship of thinness.
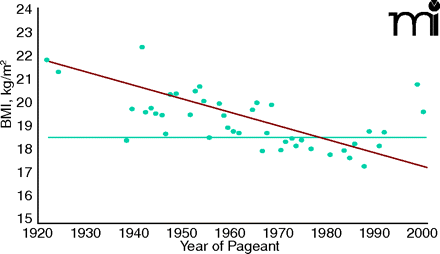
Researchers at the Johns Hopkins School of Public Health calculated the BMI of every Miss America from 1922 to 1999, using data on file at the Miss America Organization. A plot of their BMI values is shown in the figure below (17). From the 1920s to the mid-1960s the winners had BMIs between 20 and 25, within the range considered normal by the World Health Organization. Since then, the majority of winners have had BMIs lower than 18.5, considered in the range of undernutrition, with one as low as 16.9. Overall, the graph shows a continuing decline in BMI throughout the century, reflecting an ever-growing perception that thinner is better (17). This persists into the twenty-first century; the slim 19-year old beauty who was crowned Miss America 2008 is said to have once suffered from an eating disorder (18).
Before 1938, new drugs could be introduced without prior review and approval by the FDA. After 1938 proof of safety had to be submitted, but it was not until 1962 that proof of effectiveness was required. Thus, many medicaments were promoted on the basis that they ought to work, although the supporting evidence was often flawed. The metabolic stimulants are an example.
Metabolic Stimulants: Thyroid
Elucidation of the function of the thyroid gland in the late nineteenth century led to the medicinal use of dried, powdered thyroid glands of domesticated food animals. Thyroid powder is effective in treating thyroid deficiency states such as cretinism, myxedema, and goiter. Among its other effects, the thyroid raises the basal metabolic rate (BMR), which is well below normal in hypothyroidism, leading to the supposition that increased thyroid function (through thyroid powder supplementation) would “burn off” body fat in obese patients. Actually, most obese persons have normal thyroid function, and thyroid produces weight loss only at toxic doses (8). Warnings against giving powdered thyroid gland to euthyroid persons were published as early as 1921 (9). Still, its use for weight loss persisted even into the 1960s, when the FDA forced thyroid-containing combination products off the market.
Metabolic Stimulants: Dinitrophenol
A number of nitrophenols used in making dyes and explosives were found to raise body temperature and increase basal metabolism. It was therefore reasoned that the putative effect of thyroid on body weight could now be realized without resorting to its use. Choosing 2,4-dinitrophenol, Tainter and Cutting (at Stanford) carried out extensive pharmacologic studies in rats, rabbits, cats, and dogs (10), and conducted clinical tests that showed the compound raised BMR by 40%, causing subjects to lose weight without following a restricted diet. While suggesting that 2, 4-dinitrophenol might be useful in treating obesity, they warned that it should be used only experimentally, under close medical observation (11).
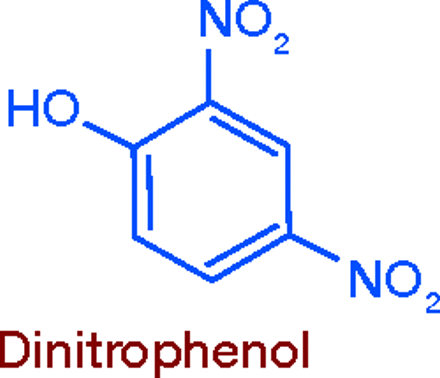
Of course, cautions were disregarded and the drug was widely introduced in 1933:by 1934, it had been given to an estimated 100,000 persons in the US. Twenty wholesalers were selling the bulk compound, and it was included in various patent medicines for self medication (11).Toxic effects, including deaths from hyperpyrexia, surfaced quickly. In 1935, several women taking the drug developed cataracts, and agranulocytosis was reported. Books and magazine articles appeared, exposing the dangers of dinitrophenol, but the FDA had no authority to act. There was a widely held view that obesity was a cosmetic issue, not a disease, and the Pure Food and Drug Act of 1906 did not give FDA jurisdiction over cosmetics. Even if dinitrophenol were admitted to be a drug, there was no law by which its use could be limited to prescription. Only after passage of The Food, Drug, and Cosmetic Act of 1938 was the FDA able to take effective action.And it did:by June 1938, dinitrophenol-containing weight reducers were gone from the market (11).
Sympathomimetic Amines: Amphetamines
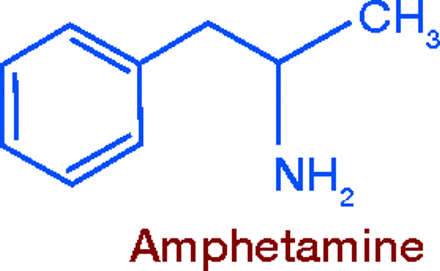
Research on compounds related to epinephrine and ephedrine led, in 1927, to the synthesis of amphetamine (Benzedrine®). A powerful central nervous system (CNS) stimulant, its sulfate salt was first used to treat narcolepsy, orthostatic hypotension, barbiturate poisoning, and other conditions. The oily, volatile base was administered via an inhaler for nasal decongestion. Amphetamine’s appetite-suppressing effect was soon recognized, and by the 1930s it was used to promote weight loss.
In addition to CNS stimulation, amphetamine and its congeners exert the peripheral actions common to the indirectly acting sympathomimetics. The site of their anorectic action is probably the lateral hypothalamic feeding center (12). The enantiomer dextroamphetamine is twice as potent as the racemic compound and was soon adopted for the same use. As a corrective against excessive CNS stimulation, the sedative amobarbital was combined with dextroamphetamine. This product, Dexamyl®, was one of the first products formulated in the earliest sustained release capsule, the Spansule®, developed in the 1950s. Methamphetamine, first synthesized in 1919, was another amphetamine derivative hailed as an appetite suppressant.
Problems with the amphetamines soon surfaced. First, tolerance to the anorectic effect almost invariably develops. Then, the use of amphetamines to ward off sleep and increase alertness and energy soon led to widespread drug abuse. Federal legislation has placed these drugs into schedule C-II, the most highly restricted category of controlled drugs (Box 3). Amphetamine barbiturate combinations were banned in the late 1960s, following the FDA’s drug efficacy review. At present, a few amphetamine products remain on the market, indicated only for narcolepsy and attention deficit hyperactivity disorder (ADHD).
Schedules of the Controlled Substances Act
SCHEDULE I
High potential for abuse
No accepted medical use in US
Not accepted as safe for use under medical supervision
SCHEDULE II
High potential for abuse
Currently accepted medical use in US
Abuse may lead to severe dependence
SCHEDULE III
Less abuse potential than Schedules I and II
Currently accepted medical use in US
Abuse may lead to low/moderate physical, or high psychological, dependence
SCHEDULE IV
Low abuse potential relative to Schedule III
Currently accepted medical use in US
Abuse may lead to limited dependence, relative to Schedule III
SCHEDULE V
Low abuse potential relative to Schedule IV
Currently accepted medical use in US
Abuse may lead to limited dependence relative to Schedule IV
Adapted from (19).
Other Sympathomimetics
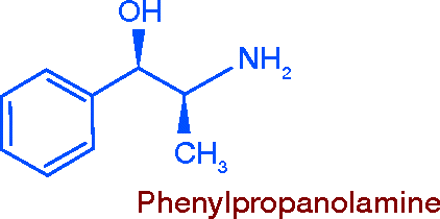
A number of chemical relatives of amphetamine, less powerful as CNS stimulants and therefore less abusable have been developed as anorectics. The oldest, first synthesized in 1912, is phenylpropanolamine (PPA). For many years PPA was employed as a nasal decongestant, and was included in a variety of cough and cold products for that purpose. Its appetite suppressant potential was discovered later, and it came to be widely promoted in the 1980s, as it had the advantage of “grandfather” status as an over-the-counter (OTC) drug. Extensive use of PPA led to a spate of serious side-effects, mainly those associated with acute hypertension (1), leading to the removal of PPA from the market.
Among the prescription-only drugs, diethylpropion, which possesses low abuse potential (Schedule C-IV), was introduced in the late 1950s. The 1960s saw the development of several more compounds: phenmetrazine (Preludin®, Schedule C-II), its derivative phendimetrazine (Plegine®, Statobex®, Schedule C-III), and phentermine (Ionamin®, Adipex-P®, Schedule C-IV). All the drugs in this group are similar in effectiveness. Long-term weight loss can be achieved only by taking them continuously. Certain issues are still unsettled, including the selection of patients most likely to benefit, whether continuous or intermittent administration is best, and optimal duration of treatment (12).
Serotonergic Agents: Fenfluramine
Fenfluramine (Pondimin®) promotes 5-hydroxytryptamine (5-HT, serotonin) release and inhibits its uptake, thereby increasing serotonergic neurotransmission, which is thought to act on the satiety center to suppress the appetite (1). In the controlled drugs schedule, fenfluramine is listed neither as a stimulant nor a depressant, but is still in the C-IV category, indicating some concern about potential abuse.
As fenfluramine is distinct from the sympathomimetics, it seemed logical to co-administer it with phentermine, probably the most benign of the sympathomimetic anorectics. This combination was designed to attack hunger by two different mechanisms and reduce the side effects of each separate drug, as lessened amounts of each drug would presumably be necessary. Thus was born “The Fen Phen Combination,” which received enormous coverage in the lay press (1). The two drugs were never combined into one product, but were taken concomitantly by the patient. Unfortunately, fenfluramine, either singly or as Fen Phen, did produce some very serious adverse effects, such as neurotoxicity, heart valve problems, and primary pulmonary hypertension (1, 12). Fenfluramine was withdrawn from the market in 1998; phentermine still remains.
Monoamine Reuptake Inhibitors: Sibutramine
Sibutramine (Meridia®) is a selective reuptake inhibitor of norepinephrine and serotonin. Originally designed as an antidepressant, it failed as such in clinical trials, but weight loss was a notable side effect. A meta-analysis of clinical studies on sibutramine ingestion demonstrated a modest degree of weight loss. Sibutramine shares many of the side effects of the other CNS-active anorectics (4). Approved in 1997, sibutramine is a Schedule C-IV drug.
Lipase Inhibitors: Orlistat
The only non-centrally acting weight loss drug, orlistat (Xenical®), exhibits a unique mode of action. Orlistat is a complex aliphatic ester that combines with both gastric and intestinal lipases, inactivating them. Thus, ingested fats are not hydrolyzed to free fatty acids but remain as non-absorbable triglycerides. A 120-mg dose of orlistat inhibits dietary fat absorption by about 25% (13). Only a small fraction of the administered dose is absorbed into the circulation. Because orlistat does not affect the neurotransmitters of the CNS, there should be no danger of abuse or addiction. On the other hand, as much of the eaten fat is excreted unchanged, gastrointestinal symptoms such as oily spotting and gas occur, as well as loose, frequent, or urgent stools (14).
Prescription use of orlistat received FDA approval in April 1999, and in February 2007 a 60-mg (half-strength) capsule was approved for OTC use under the name Alli®. It is marketed in a rather elaborate way, in order to reach out to the user. A “starter pack” includes a carrying case that holds one day’s dosage, room for a multivitamin, and six brochures to help patients with their weight loss plan. Web site resource guides are also available for patients and health care professionals (14). It will be interesting to see if Alli® becomes a blockbuster product, as expected, or if some problem will surface, as has been the case with the systemically acting weight loss medications.
Conclusion
It is hardly possible to fully characterize the attitudes of the American people; the population is so large, diverse, and non-homogeneous. Yet, two opposing trends seem to stand out through the past one hundred years. Eating patterns have evolved that favor the over-consumption of fats and carbohydrates. Additionally, especially during the past six decades, technology has freed most American men and women from hard physical labor, and fostered spectator, rather than participatory, sport and entertainment. Together, these factors have produced rising rates of overweightness and obesity. At the same time, there has been an ongoing preoccupation with weight loss and achieving a slim body image. Toward this end, one drug modality after another has been adopted, with little success, and often with unfortunate results. Outside of surgery (i.e., stomach “stapling” and liposuction), there appears no better method to achieve weight reduction and better health than by reducing one’s caloric intake and increasing one’s physical activity.
- © American Society for Pharmacology and Experimental Theraputics 2008
References

Stanley Scheindlin, DSc, holds a BS in pharmacy from Temple University and graduate degrees in pharmaceutical chemistry from Philadelphia College of Pharmacy and Science (now University of Sciences in Philadelphia). His academic research dealt with plant constituents and chemical interactions of vitamins. In his pharmaceutical industry career, he handled new drug formulation developments, and later regulatory affairs, presiding over the filing of about 100 generic new drug applications and two innovative drug applications. Now retired, his activities include volunteer work, consulting, and writing Reflections pieces for this journal.






