|
 
 |
| ORIGINAL ARTICLE |
|
| Year : 2013 | Volume
: 1
| Issue : 1 | Page : 11-16 |
|
Effects of different concentrations of hydrogen peroxide on the color stability of various esthetic restorative materials in vitro
Haluk Baris Kara1, Tevfik Yavuz2, Ali Riza Tuncdemir3, Ozgun Yusuf Ozyilmaz2
1 Department of Prosthodontics, Faculty of Dentistry, Istanbul Medipol University, Istanbul, Turkey
2 Department of Prosthodontics, Faculty of Dentistry, Selcuk University, Konya, Turkey
3 Department of Prosthodontics, Faculty of Dentistry, Konya Necmettin Erbakan University Faculty of Dentistry, Konya, Turkey
| Date of Web Publication | 16-Aug-2013 |
Correspondence Address:
Haluk Baris Kara
Istanbul Medipol University, Ataturkbulv. No. 27 unkapani, 34083 Fatih, Istanbul
Turkey
 Source of Support: None, Conflict of Interest: None  | Check |
DOI: 10.4103/2347-4610.116587

Purpose: The aim of this in vitro study was to determine the color changes of four different restorative materials after exposing these materials to two different bleaching agents. Materials and Methods: In this study, bleaching agents were applied to a low-fusing porcelain (VITA VM9), heat-pressed glass ceramics (IPS Empress Esthetic), and two types of composites (Clearfil Majesty Esthetic and Clearfil Photo Posterior). Twenty disc-shaped specimens were fabricated (with a diameter of 10 mm and a thickness of 2 mm) from each material (n0 = 10). The initial color changes were measured using a spectrophotometer. The first set of specimens were bleached with 10% hydrogen peroxide (HP) for 1 h daily for 10 days. Another set of specimens were bleached with 40% HP bleaching gel for two consecutive applications for 20 min each. The data were analyzed statistically by using two-way analysis of variance (ANOVA) and post hoc Tamhane's T2 tests ( P < 0.05). Results: The mean color change value (ΔE) for Clearfil Majesty Esthetic was significantly higher than the value for VITA VM9 groups, treated with the 10% HP. In addition, the values for Clearfil Photo Posterior were significantly higher than the values for Clearfil Majesty Esthetic, IPS Empress Esthetic, and VITA VM9 groups treated with the 40% HP (P < 0.05). The comparative evaluation of bleaching agents on the color change of materials revealed that the differences between the bleaching agents were significant for the Clearfil Photo Posterior groups (P < 0.05). Conclusion: The effects of bleaching agents should be known and applied consciously when restorative materials (especially polymer-containing restorative materials) are present. Keywords: Bleaching, color, dental materials
How to cite this article:
Kara HB, Yavuz T, Tuncdemir AR, Ozyilmaz OY. Effects of different concentrations of hydrogen peroxide on the color stability of various esthetic restorative materials in vitro. Eur J Prosthodont 2013;1:11-6 |
How to cite this URL:
Kara HB, Yavuz T, Tuncdemir AR, Ozyilmaz OY. Effects of different concentrations of hydrogen peroxide on the color stability of various esthetic restorative materials in vitro. Eur J Prosthodont [serial online] 2013 [cited 2018 Jul 12];1:11-6. Available from: http://www.eurjprosthodont.org/text.asp?2013/1/1/11/116587 |
| Introduction | |  |
The number of people seeking optimum dental esthetics is growing worldwide, and one of the most common esthetic problems that patients hope to rectify is discolored teeth. [1] Teeth can be treated using various restorative techniques, such as direct composite veneers, indirect porcelain veneers, ceramic crowns, or even bleaching. [2] Although new esthetic restorative materials are being introduced almost daily, porcelain remains as the esthetic material of choice for most clinicians. Preference for porcelain restorations, in many forms, arises from their excellent biocompatibility, their strength and surface texture of the material, and their overall esthetic properties. [3]
Using bleaching techniques to improve the esthetics of the natural dentition has been becoming increasingly popular since 1989. [4] Patients' interest in cosmetic dentistry has contributed to the development of new bleaching materials and techniques. [5] Bleaching is a relatively noninvasive approach to lightening teeth that is stained extrinsically or intrinsically. Bleaching techniques may be classified by whether they involve vital or nonvital teeth or whether the procedure is performed in-office or has an at-home component. [2],[6],[7] In-office bleaching is administered by a dentist and staff members using higher concentrations of whitening agents, and at-home bleaching is administered by the patient, using lower concentrations of whitening agents in special trays. [8],[9]
The in-office technique uses bleaching agents, containing high concentrations of carbamide peroxide (35-37%) [10],[11],[12] or hydrogen peroxide (HP; 30-46%). [13],[14],[15] The home-use dental bleaching systems contain various concentrations of carbamide peroxide, ranging between 10% and 22%, [9],[11],[14] and HP between 3% and 10%. [9],[15]
New home bleaching agents also continue to enter the market, and they vary with respect to peroxide concentration, delivery method, contact time, product formulation, and other factors, all of which have the potential to impact the individual's overall clinical tolerability and the individual's oral response. [9],[16] However, patients need to be informed of bleaching's potential effects on the structure and color of enamel and restorative materials. These new products, combined with consumer's greater cosmetic awareness and desire, lead to a need for controlled, randomized, nonbiased comparisons of available home bleaching products. Such research studies provide one source for a comparative assessment of the safety and effectiveness of the various vital bleaching systems. Color research is particularly amenable to comparative testing, because objective measurement methods for color are readily available. [16] Numerous studies have shown that bleaching is effective at whitening certain types of discolored teeth, but its effect on restorative materials is not clearly understood. [17],[18],[19],[20],[21],[22]
Therefore, the purpose of this study was to determine the quantitative color changes in four different restorative materials as a result of simulated bleaching with 40% and 10% HP in vitro. The research hypothesis was that both bleaching systems would have significant effects on the color change of restorative materials.
| Materials and Methods | |  |
This study tested two bleaching products on four restorative materials. The materials, product names, and manufacturers are listed in [Table 1].
A leucite-based core (IPS Empress Esthetic, Ivoclar Vivadent Schaan, Liechtenstein) specimens (10 mm in diameter and 2 mm in thickness) were waxed (BEGO, Bremen, Germany), sprued, and then pressed after investment. All procedures were performed with IPS Empress Esthetic materials. For the fabrication of feldspathic ceramic discs (VITA VM 9, Vita Zahnfabrik Bad Säckingen, Germany), a mold was made using vinyl polysiloxane putty (Virtual, Ivoclar Vivadent Schaan, Liechtenstein) to facilitate the fabrication of the porcelain discs (10 mm in diameter, 2 mm thick). The porcelain was mixed with sculpting liquid and condensed into the mold. Tissue (Selpak, Eczacibasi Holding, Istanbul, Turkey) was used to absorb the excess moisture. After drying, the discs were carefully removed from the mold, placed on a sagger tray, and fired according to the manufacturers' recommendations (950°C) in a porcelain oven (Vita Vacumat 40 T, Vita Zahnfabrik). Forty discs (20 for each porcelain tested) were made. The color A2 or the color corresponding to A2 was selected for each material. The specimens were then trimmed with a thin, cylindrical diamond bur (D-Z Labor, Drendel and Zweiling GmbH and Co., Berlin, Germany) and were exposed to air abrasion with 50-μm aluminum-oxide powders. All the ceramic specimen surfaces were then polished with a special polishing kit (Optrafine, Ivoclar, Schaan, Liechtenstein) that had a slow-speed handpiece (NSK, Tokyo, Japan) running at 15,000 rpm.
The holes, 10 mm in diameter, were drilled in a 2-mm-thick polytetrafluoroethylene plate to form the composite (Clearfil Majesty Esthetic and Clearfil Photo Posterior) specimens. These restorative materials were placed into the mold separately and sandwiched between two glass plates. In accordance with the manufacturer's directions, a curing light (800 mW/cm 2 ) was applied to the top of the filled molds for 40 s by a light-polymerizing unit (Bluephase, Ivoclar Vivadent Schaan, Liechtenstein). The distance between the light source and the specimen was standardized using of a 1-mm glass slide. Forty composite specimens were made for this study. The specimens were polished with medium, fine, and superfine polishing kits (Astrapol, Ivoclar Vivadent Schaan, Liechtenstein) on a slow-speed handpiece (10,000 rpm) in accordance with the manufacturer's directions.
After the finishing procedures, specimens were subjected to ultrasonic treatment (Biosonic UC 50, Coltene Whaledent, Cuyahoga Falls, OH, USA) in distilled water to remove any surface residues. They were then dried. All specimens were stored in distilled water in screw-top vials (Isolab, Laborgeräte GmbH, Wertheim, Germany) at room temperature for 24 h before any testing procedure.
Eighty specimens were randomly divided into two groups (n = 10) according to the bleaching procedure. The first group specimens were bleached with the bleaching protocol simulated a typical in-office whitening procedure. Two consecutive applications of a 40% HP gel (Opalescence Boost; Ultradent Products, Inc., South Jordan, Utah, USA), for 20 min each, were done. Custom bleaching trays were made for each block from a flexible plastic vacuum-formed material (Sof-Tray sheets, Ultradent Products, Inc.) that fully covered the block, but left solid pillars of plastic (1 mm in height) to act as spacers. This configuration provided a consistent gel thickness. Used bleach was removed and replaced by new material during the successive applications. The treated plates were rinsed with water and returned to their individual storage tubs. The second group specimens were bleached with the bleaching protocol and simulated a typical home whitening procedure. For this treatment, the flexible vacuum-formed material was filled with HP gel (Opalescence Trèswhite Supreme 10%; Ultradent Products, Inc.) for 1 h. During bleaching, the block with a bleaching tray was placed into an individual, sealed storage container and placed in a 37°C oven. At the end of the 1-h bleaching period, the bleach was removed and the block was returned to its water storage container. This process was repeated for 10 consecutive days.
Color measurement
Before and after the treatment, the color of every specimen was measured with a spectrophotometer (VITA Easyshade, VITA Zahnfabrik). This instrument measures the spectral reflectance of a color and converts it into a tristimulus value, which is the internationally accepted numerical form. The spectrophotometer's CIE L*a*b* output is based on D65 illuminant and a 2° standard observer. Three measurements were made, and the average reading was calculated for each specimen. The instrument was recalibrated after measurement of each group (n = 10). The CIE L*a*b* measurements make it possible to evaluate the amount of perceptible color change in each specimen. The CIE L*a*b* color space is a uniform three-dimensional color order system. Equal changes in any of the three coordinates can be perceived as visually similar. Total color differences were calculated with the following equation: [23],[24]
ΔE*=[(ΔL*) 2 +(Δa *) 2 +(Δb *) 2 ] 1/2
The critical remarks of the color change (ΔE ) were quantified by the National Bureau of Standards (NBS), which rates the way that a color change is evaluated by the human eye [Table 2]. The formula used for this conversion is NBS units = ΔE × 0.92.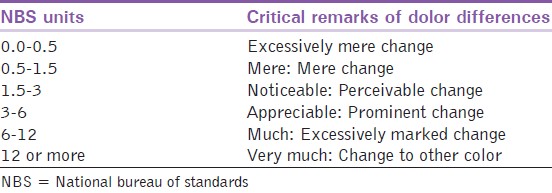 | Table 2: National bureau of standards system of expressing color differences
Click here to view |
Statistical analysis
The results of scores were entered into a spreadsheet (Excel, Microsoft, Redmond, WA, USA) for calculation of descriptive statistics. Statistical analyses were performed with SPSS 20 (Windows; SPSS Inc., Chicago, IL, USA) for WINDOWS (α = 0.05). The data of color differences were statistically analyzed with two-way ANOVA, to evaluate the performances of the different bleaching technique and the interaction between bleaching technique and the restorative materials. The homogenity of variances was measured by using Levene's test. Tamhane's T2 tests for post hoc analysis were used for continuous variables normally distributed with unequal variances. P values less than 0.05 were considered to be statistically significant in all tests (P < 0.05) .
| Results | |  |
The means of color change (ΔE) along with standard deviations (SD) of the four different restorative materials after bleaching are presented in [Table 3]. Statistically significant color changes were found in both bleaching agent groups (P < 0.05) [Table 4]. The Tamhane multiple comparison test showed that the mean color change value (ΔE) for Clearfil Majesty Esthetic was significantly higher than the values for VITA VM9 (P < 0.05); there was no significant difference in the color change values among Clearfil Majesty Esthetic, IPS Empress Esthetic, and Clearfil Photo Posterior (P > 0.05), groups treated with the 10% HP. In addition, the values for Clearfil Photo Posterior was significantly higher than the values for Clearfil Majesty Esthetic, IPS Empress Esthetic, and VITA VM9 (P < 0.05). Furthermore, there was no significant difference in color change values among Clearfil Majesty Esthetic, IPS Empress Esthetic, and VITA VM9 (P > 0.05), groups treated with the 40% HP.
The comparative evaluation of bleaching agents on the color change of materials revealed that the differences between the bleaching agents were significant for the Clearfil Photo Posterior groups (P < 0.05).
| Discussion | |  |
Color changes can be evaluated using a visual method and color measurement devices. Most of the color measurement devices utilized in dentistry use the ΔE from the Commission International de I'Eclairage CIE (L*a*b*) color system to determine the color differences or changes. In this scheme, color is measured in three coordinate dimensions of L* that represents lightness (from white to black; similar to value) a* corresponds to the green-red axis (the negative value indicates green; the positive value indicates red), and b* corresponds to the blue-yellow axis (the negative value indicates blue; the positive value indicates yellow). [25]
Colorimeters and spectrophotometers are used for color analysis in dental research. The literature has shown that there are no significant differences between colorimeter and spectrophotometer measurements. [26],[27],[28] The most important differences between colorimeter and spectrophotometer measurements are their wavelength range and the diversity of the light sources they use. In the clinic, the numerical values of minimal color changes and differences are not possible to distinguish visually. Results vary over time from person to person and for the same people as well. Statistically available and objectively evaluated results of color can be obtained from color-measuring devices like the spectrophotometer. [29] For these reasons, in this study, the spectrophotometer was used to detect fine color changes.
Color measurement studies always make more than one measurement for each specimen. Based on measuring the various regions of each specimen, average values were obtained in one study through five color measurements per specimen, [29] and other studies have made three measurements per specimen. [30],[31] This study copies these latter studies and took three measurements of each sample (looking at three different regions on each sample), and then averaged these three values for each sample.
Multiple measurements are important because the same sample can have varying thicknesses, which will affect the darkness or lightness of one region's color. One study found that thickness and light transmission affect the color of a sample. [32] In response to this finding, previous color studies [33],[34],[35] standardized all samples to be 2 mm in thickness. Other previous studies prepared the color samples to be 10 mm in diameter [36],[37] and 6 mm in diameter, [29] which is suitable for the optical screen of the color measurement device.
This in vitro study measured the quantitative color changes of four different restorative materials as a result of simulated bleaching with 10% and 40% HP. The results of this study support the hypothesis that the different bleaching systems produce different results on restorative materials because there were significant differences in color change within groups.
After implementing 10% or 40% HP on the materials (a heat-pressed glass ceramics, a low-fusing porcelain, and two types of composites), the color changes in the materials were examined. Color change (ΔE) values that were found in the restorative materials were as follows: After application of 10% HP-Clearfil Majesty Esthetic = 2.63, Clearfil Photo Posterior = 2.37, IPS Empress Esthetic = 1.59, and VITA VM9 = 1.07; and after the application of 40% HP-Clearfil Majesty Esthetic = 2.37, Clearfil Photo Posterior = 5.08, IPS Empress Esthetic = 1.58, and VITA VM9 = 0.94. For NBS unit color evaluation, the results between the values of 0.5 and 1.5 are "mere," results, between the values of 1.5 and 3 are "noticeable," and results between 3 and 6 are "appreciable" color changes. [30] The restorative materials (Clearfil Majesty Esthetic, Clearfil Photo Posterior, and IPS Empress Esthetic) exhibited "noticeable" changes in color after 10% HP implementation. The 10% HP-treated VITA VM9 porcelain showed a "mere" color change. The Clearfil Majesty Esthetic and IPS Empress Esthetic treated with 40% HP underwent "noticeable" changes. VITA VM9 porcelain showed a "mere" color change, and Clearfil Photo Posterior showed "appreciable" changes after 40% HP treatment.
In this study, however, the heavy-filled hybrid composite resin Clearfil Photo Posterior exhibited the highest mean values color change (ΔE), which seems to be the result of the highest filler load level (86 wt%) and the largest average particle size (4 μm) of the material than the other composite material used in the study. Also, it is known that the mechanical properties of the materials with a polymer structure, such as composite materials, are reduced due to their ability to absorb water. [38] It is thought that the materials with a polymer structure have less color stability than the porcelains, when exposed to several chemical agents. The result of this study indicated that the color change induced by the HP agent might be dependent on the matrix structure, filler volume, as well as the filler type of different types of resin-based composites tested. This finding is in agreement with the results of Hubbezoglu et al. [39]
Canay and Cehreli [30] have also observed that as a result of the application of a 10% carbamide peroxide bleaching agent, the change in color was greater in polyacid-modified composites than in composites with hybrid and macrofilling. They have reported that the change in color is associated with the matrix content, the amount of filler, and the composite type.
Several previous studies have reported that there are not any clinical and statistical differences in the color of glazed porcelain restoration materials treated with bleaching agents. [40] In a clinical study, Haywood [4] reported that the "nightguard vital bleaching" method, which contains 10% carbamide peroxide, had no effect on glazed porcelain and that the discoloration that occurs in porcelain restorations, which is not reinforced with metal, is connected to the change in the color underneath the tooth's surface. Another study has reported that the bleaching process applied to the vital teeth had no effects on the color change or other physical properties of porcelain restoration materials. [41] The results of previous studies are different from those of this study, and the reasons behind this are presumably that the restorative materials were different and that the porcelain sample surfaces were not glazed in the present study.
In a study by Kao and others, [42] the effects of the bleaching agents containing 10% carbamide peroxide on dental structure and on feldspathic porcelain (Finesse) restorative material were examined. Although the discoloration was less noticeable on the porcelain than it was on the dental structures, the ΔE value was determined to be 1.2. This result agrees with the current study.
In a study conducted by Zaki and Fahmy, [22] it was reported that the bleaching agent containing 15% carbamide peroxide had numerically little whitening effect on autoglazed feldspathic porcelain (Duraceram) because it did not bring about any statistically significant change in color. However, it has been observed that discoloration occurs on the surfaces of feldspathic porcelain treated with overglaze and polishing, as a result of the application of a bleaching agent. Similarly, in a study by Rosentritt and others, [43] the bleaching agent was reported to bring about a change in color on some restorative materials (composite, compomer, and ormocer). Using a spectrophotometer, Li and colleagues [44] found significant changes in the color of nanohybrid and packable composite resins after bleaching with 15% carbamide peroxide. These results also support the results of the current study.
There are some limitations of the current study design that must be noted. As in many in vitro studies, the oral environment cannot be fully simulated. It would be useful to support the results of this study with other clinical studies or with the studies that could imitate the oral environment better in order to find out whether the effects of the bleaching agents change with exposure to saliva. The spectrophotometer was used to detect fine color changes in this study. However, in further studies other analysis methods (colorimeters or even digital cameras and imaging systems) can be used.
| Conclusion | |  |
Within the limitations of this study and regarding the spectrophotometric evaluation, it was concluded that statistically significant differences, among the color changes of all test groups, were found after exposing them to bleaching agents. Dentists should make sure that their patients with dental restorations (especially those with polymer content) are aware of the changes that may occur during whitening, as well as the possibility that their bleached restorations may need to be polished or replaced at the end of the treatment.
| References | |  |
| 1. | Turkun LS, Turkun M. Effect of bleaching and repolishing procedures on coffee and tea stain removal from three anterior composite veneering materials. J Esthet Restor Dent 2004;16:290-301. 
|
| 2. | Polydorou O, Monting JS, Hellwig E, Auschill TM. Effect of in-office tooth bleaching on the microhardness of six dental esthetic restorative materials. Dent Mater 2007;23:153-8. 
|
| 3. | Ertan AA, Sahin E. Colour stability of low fusing porcelains: An in vitro study. J Oral Rehabil 2005;32:358-61. 
[PUBMED] |
| 4. | Haywood VB, Heymann HO. Nightguard vital bleaching. Quintessence Int 1989;20:173-6. 
[PUBMED] |
| 5. | Haywood VB. Achieving, maintaining and recovering successful tooth bleaching. J Esthet Restor Dent 1996;8:31-8. 
|
| 6. | Rodrigues JA, Marchi GM, Ambrosano GM, Heymann HO, Pimenta LA. Microhardness evaluation of in situ vital bleaching on human dental enamel using a novel study design. Dent Mater 2005;21:1059-67. 
[PUBMED] |
| 7. | Taher NM. The effect of bleaching agents on the surface hardness of tooth colored restorative materials. J Contemp Dent Pract 2005;6:18-26. 
[PUBMED] |
| 8. | Luca G, Fabio B, Claudia B, Michele N, Daniele SR. A randomized clinical trial comparing at-home and in-office tooth whitening techniques. J Am Dent Assoc 2010;141:1357-64. 
|
| 9. | Matis BA, Mousa HN, Cochran MA, Eckert GJ. Clinical evaluation of bleaching agents of different concentrations. Quintessence Int 2000;31:303-10. 
[PUBMED] |
| 10. | Sulieman M. An overview of bleaching techniques: 2. Night guard vital bleaching and non-vital bleaching. SADJ 2006;61:352-4. 
[PUBMED] |
| 11. | Wattanapayungkul P, Yap AU. Effects of inin-officebleaching products on surface finish of tooth-colored restorations. Oper Dent 2003;28:15-9. 
[PUBMED] |
| 12. | Swift EJ Jr, Perdigao J. Effects of bleaching on teeth and restorations. Compend Contin Educ Dent 1998;19:815-20. 
|
| 13. | Marshall MV, Gragg PP, Packman EW, Wright PB, Cancro LP. Hydrogen peroxide decomposition in the oral cavity. Am J Dent 2001;14:39-45. 
[PUBMED] |
| 14. | Campos I, Briso AL, Pimenta LA, Ambrosano G. Effects of bleaching with carbamide peroxide gels on microhardness of restoration materials. J Esthet Restor Dent 2003;15:175-83. 
[PUBMED] |
| 15. | Atali PY, Topbasi FB. The effect of different bleaching methods on the surface roughness and hardness of resin composites. J Dent Oral Hyg 2011;3:10-1. 
|
| 16. | Karpinia K, Magnusson I, Barker ML, Gerlach RW. Clinical comparison of two self-directed bleaching systems. J Prosthodont 2003;12:242-8. 
[PUBMED] |
| 17. | Cabrika RM, Myers M, Downey MC. Clinical study of tooth shade. Lightening from dentist-suervised, patientapplied treatment with 10% carbamide peroxide gels. J Esthet Dent 1999;11:325-31. 
|
| 18. | Oltu U, Gurgan S. Effects of three concentrations of carbamide peroxide on the structure of enamel. J Oral Rehabil 2000;27:332-40. 
|
| 19. | Metzler KT, Woody RD, Miller AW 3 rd , Miller BH. In vitro investigation of the wear of human enamel by dental porcelain. J Prosthet Dent 1999;81:356-64. 
|
| 20. | White DJ, Kozak KM, Zoladz JR, Duschner HJ, Goetz H. Impact of crest night effects bleaching gel on dental enamel, dentin and key restorative materials. In vitro studies. Am J Dent 2003;16:22-7. 
|
| 21. | Attin T, Hannig C, Wiegand A, Attin R. Effect of bleaching on restorative materials and restorations a systematic review. Dent Mater 2004;20:852-61. 
[PUBMED] |
| 22. | Zaki AA, Fahmy NZ. The effect of a bleaching system on properties relatedto different ceramic surface textures. J Prosthodont 2009;18:223-9. 
[PUBMED] |
| 23. | Knispel G. Factors affecting the process of color matching restorative materials to natural teeth. Quintessence Int 1991;22:525-31. 
[PUBMED] |
| 24. | Ozturk O, Uludag B, Usumez A, Sahin V, Celik G. The effect of ceramic thickness and number of firings on the color of two all-ceramic systems. J Prosthet Dent 2008;100:99-106. 
[PUBMED] |
| 25. | Chu SJ, Devigus A, Mieleszko A. Fundamentals of color shade matching and communication in esthetic dentistry. 1 st ed. St. Louis: Quintessence Publication Books; 2004. p. 85-8. 
|
| 26. | Seghi RR. Effects of instrument measuring geometry on colorimetric assessments of dental porcelains. J Dent Res 1990;69:1180-3. 
[PUBMED] |
| 27. | Burgt TP, Bosch JJ, Borsboom PC, Kortsmit JP. A comparison of new and conventional methods for quantification of tooth color. J Prosthet Dent 1990;63:155-62. 
|
| 28. | Goldstein RE, Schmitt GW. Repeatibility of a designed intraoral colorimeter. J Prosthet Dent 1993;69:616-9. 
|
| 29. | Kolbeck C, Rosentritt M, Behr M, Lang R, Handel G. Discoloration of facing and restorative composites by UV irradiation and staining food. Dent Mater 2006;22:63-8. 
|
| 30. | Canay S, Cehreli MC. The effect of current bleaching agents on the color of light-polymerized composites in vitro. J Prosthet Dent 2003;89:474-8. 
[PUBMED] |
| 31. | Bailey SJ, Swift EJ. Effects of home bleaching products on composite resins. Quintessence Int 1992;23:489-94. 
|
| 32. | Arikawa H, Fuji K, Kanie T, Inoue K. Light transmittance characteristics of light-cured composite resins. Dent Mater 1998;14:405-11. 
|
| 33. | Doray PG, Wang X, Powers JM, Burgess O. Accelerated aging effects color stability of provisional restorative materials. J Prosthodont 1997;6:183-8. 
|
| 34. | Ikeda T, Sýdhu SK, Omata Y, Fujita M, Sano H. Colour and translucency of opaque-shades and body-shades of resin composites. Eur J Oral Sci 2005;113:170-3. 
|
| 35. | Sampath S, Ramachandra GS. Effects of glass fibers on light transmittance and color of fiber-reinforced composites. Dent Mater 2008;24:211-5. 
|
| 36. | Douglas RD. Color stability of new generation indirect resins for prosthodontic application. J Prosthet Dent 2000;83:166-70. 
[PUBMED] |
| 37. | Nakamura T, Saito O, Mizuno M, Tanaka H. Changes in translucency and color of particulate filler composite resins. Int J Prosthodont 2002;15:494-9. 
[PUBMED] |
| 38. | Lassila LV, Nohrström T, Vallittu PK. The influence of short-term water storage on the flexural properties of unidirectional glass fiber-reinforced composites. Biomaterials 2002;23:2221-9. 
|
| 39. | Hubbezoglu I, Akaoglu B, Dogan A, Keskin S, Bolayir G, Ozcelik S, et al. Effect of bleaching on color change and refractive index of dental composite resins. Dent Mater J 2008;27:105-16. 
|
| 40. | Haywood VB. History, safety, and effectiveness of current bleaching techniques and application of the nightguard vital bleaching technique.Quintessence Int 1992;23:471-88. 
[PUBMED] |
| 41. | Swift EJ Jr. Restorative considerations with vital tooth bleaching. J Am Dent Assoc 1997;128(Suppl 4):60-4. 
|
| 42. | Kao EC, Peng P, Johnson WM. Color changes of teeth and restorative materials exposed to bleaching. J Dent Res 1991;70:570. 
|
| 43. | Rosentritt M, Lang R, Plein T, Behr M, Handel G. Discoloration of restorative materials after bleaching application.Quintessence Int 2005;36:33-9. 
[PUBMED] |
| 44. | Li Q, Yu H, Wang Y. Colour and surface analysis of carbamide peroxide bleaching effects on the dental restorative materials in situ. J Dent 2009;37:348-56. 
[PUBMED] |
[Table 1], [Table 2], [Table 3], [Table 4]
|