|
 
 |
|
CASE REPORT |
|
|
|
| Year : 2011 | Volume
: 17
| Issue : 2 | Page : 100-103 |
| |
Deletion of ABL/BCR on der(9) associated with severe basophilia
Shantashri Vaidya, Manisha Madkaikar, Kanjaksha Ghosh, Babu Rao Vundinti
Department of Cytogenetics, National Institute of Immunohaematology (ICMR), K.E.M. Hospital Campus, Parel, Mumbai, India
| Date of Web Publication | 17-Oct-2011 |
Correspondence Address:
Babu Rao Vundinti
Department of Cytogenetics, National Institute of Immunohaematology (ICMR), 13th Floor, K.E.M. Hospital Campus, Parel, Mumbai - 400 012
India
 Source of Support: None, Conflict of Interest: None
DOI: 10.4103/0971-6866.86198

 Abstract Abstract | | |
Chronic basophilic leukemia is a rare form in chronic myeloid leukemia patients. Only limited number of reports are available. Herein, we describe a patient who presented with fatigue, weight loss, leucocytosis, prominent basophilia, and mild eosinophilia. On biopsy, bone marrow was hypercellular with marked basophils. The immunophenotype showed abnormal expression of CD7, which is suggestive of basophilic maturation. Chromosomal analysis from GTG-banded metaphases revealed Ph positivity, and fluorescence in situ hybridization (FISH) with BCR/ABL dual color, dual fusion probe showed single fusion on the der(22) chromosome and ABL/BCR fusion was deleted on the der(9) chromosome. The deletion (ABL/BCR) on der(9) may be associated with basophilia which may be also indicative of the transformation of CML to acute myeloid leukemia.
Keywords: Basophilia, BCR/ABL deletions, chronic myeloid leukemia
How to cite this article:
Vaidya S, Madkaikar M, Ghosh K, Vundinti BR. Deletion of ABL/BCR on der(9) associated with severe basophilia. Indian J Hum Genet 2011;17:100-3 |
How to cite this URL:
Vaidya S, Madkaikar M, Ghosh K, Vundinti BR. Deletion of ABL/BCR on der(9) associated with severe basophilia. Indian J Hum Genet [serial online] 2011 [cited 2016 May 13];17:100-3. Available from: http://www.ijhg.com/text.asp?2011/17/2/100/86198 |
 Introduction Introduction | |  |
Chronic myelogenous leukemia (CML) is one of the most common myeloproliferative neoplasms (MPNs) associated with basophilia and the presence of 20% basophilis or more is a characteristic feature of CML in accelerated or blast phase. [1],[2] CML patients with a poorly defined entity, Ph-/BCR/ABL-atypical CML, have a significantly lower incidence of basophilia as compared with Ph+/BCR/ABL+ CML. [3],[4] The patient we describe presented with fatigue, weight loss, leukocytosis and persistently prominent basophilia. We performed morphological examination, flow cytometry (FCM), immunophenotyping, and cytogenetic analysis to characterize chronic basophilic leukemia (CBL).
 Case Report Case Report | |  |
A 45-year-old male presented with fever, fatigue, pallor, and weight loss. A complete blood count revealed anemia (hemoglobin 5.6 g/L; mean corpuscular volume [MCV] 94.2 fL), platelet count 84 × 10 9 /L, white blood count [WBC] 173 × 10 9 /L). A differential count showed 1% neutrophils, 4% basophils, 3% eosinophils, and lymphocytes 8%. Physical examination revealed splenomegaly (3-4 cm below the left costal margin) with hepatomegaly and lymphadenopathy (tiny axillary lymph nodes). The patient had no complaints of hypertension, infection, etc.
Morphological findings
The peripheral blood (PB) examination showed leukocytosis with marked basophilia (40%) and moderate eosinophilia (17%). The bone marrow (BM) biopsy showed a markedly hypercellular (98%) marrow with myeloid hyperplasia (the myeloid to erythroid ratio was increased at 7:1), increased eosinophils, increased megakaryocytes with many small and hypolobated forms [Figure 1], and moderate reticulin fibrosis.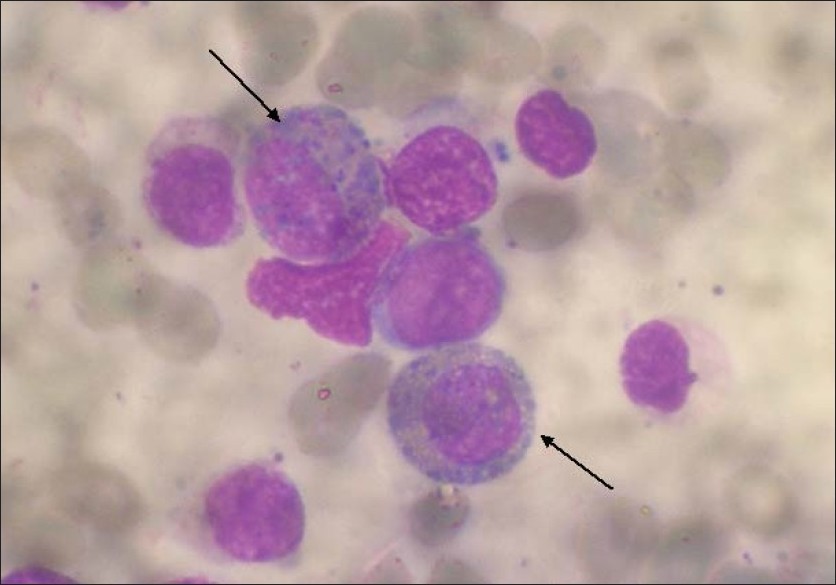 | Figure 1: Bone marrow aspirate smear stained with the Giemsa stain shows the presence of blasts small to moderate in size with two to three nucleoli and scanty cytoplasm. A marked increase in immature eosinobasophilic cells is also seen
Click here to view |
The BM aspirate showed numerous abnormal appearing basophils accounting for 66% of total nucleated cells. The cells exhibited basophilic cytoplasm, cytoplasmic hypogranulation or agranulation, and nuclear hypersegmentation. Eosinophils were 9% with abnormal granulation and nuclear hyperlobation. The blasts were 8%, dyserythropoiesis and dysgranulopoiesis. Immunohistochemical stains using CD117 and mast cell tryptase were performed on the BM core biopsy and did not reveal increased mast cells or mast cell aggregates.
FCM analysis and immunophenotype
The immunophenotype analysis was performed on the BM sample by flowcytometry using a FACS caliber instrument (Becton Dikinson Biosciences, San Jose, CA, USA). Antibodies against the following antigens were used: CD2, CD3, CD4, CD5, CD7, CD8, CD10, CD19, CD20, CD22, HLA-DR, CD11b, CD13, CD14, CD33, CD117, CD34, and CD123. The immunophenotype of the patient showed expression of CD7, HLA-DR, CD11b, CD13, CD33, CD34, CD117, CD123, and HLA-DR with aberrant expression of CD7 which is suggestive of basophilic maturation.
Cytogenetic analysis
The chromosomal preparation obtained from the BM sample using a direct method and unstimulated 24-h culture. The chromosomal preparation was subjected to standard GTG-banding and karyotyped according to ISCN 2005. The chromosomal analysis revealed 46,XY,t(9;22)(q34;q11) in 15 out of 25 metaphases scored [Figure 2]a. Metaphase fluorescence in situ hybridization (FISH) with a BCR/ABL dual fusion probe (Vysis Inc., Abbott Molecular Diagnostics, Abbott Park, IL, USA) on abnormal metaphase cells showed the BCR/ABL fusion on chromosome 22 and ABL/BCR fusion was deleted on the der(9) chromosome [Figure 2]b. As the patient exhibited Ph positivity and BCR/ABL fusion, the patient was treated with Gleevec. His WBC count did decline little, but basophilia persisted (40%) after 1-month treatment. The patient required aggressive therapy and long-term monitoring to maintain the complete blood count. | Figure 2: (a) Partial metaphase showing Ph? chromosomes der(22) and der(9). (b) FISH with the dual fusion probe showing BCR/ABL fusion on chromosome 22 and the absence of ABL/BCR on the der(9) chromosome
Click here to view |
 Discussion Discussion | |  |
The CBL was first reported by Kyle and Pease. [5] Later on others also reported similar cases. [6] While basophilia is observed with various myeloid disorders, it is considered a poor prognostic feature in the chronic phase. [1],[7] In addition, the basophilia differentiation of immature blasts may be observed in the blastic phase of CML. [8] Acute basophilic leukemia, which has been included in the revised WHO often represents the blastic transformation of CML. In our case the patient had splenomegaly and hypercellular BM with an elevated myeloid/erythroid ratio which was suggestive of CML. The immunophenotyping is important for the accurate diagnosis as morphologically the neoplastic basophils may not be easily recognized on smear because of the morphologically atypical and decreased granules. In our case, the initial automatic differential did not characterize the cells as basophils but flagged them as immature eosino-basophilic cells. FMC immunophenotyping became particularly important in confirming the nature of these cells because normal basophils characteristically display CD123+, CD22, HLA-DR and CD38+ immunophenotype. [9] The aberrant immunophenotype expression of CD64 and under expression of CD38 and CD123 were reported in CML patients with basophils. [9] However, recently the normal expression of CD45, CD123, CD13, CD33, CD119 and CD11b in a case of CBL were reported. [10]
Our case showed negative expression of CD2, CD3, CD4, CD5, CD8, CD10, CD19, CD20, CD22, CD14, and CD38. Interestingly, the blast population was positive for CD11b, CD13, CD33, CD34, CD117, CD123, and HLA-DR with aberrant expression of CD7 which suggested basophilic maturation. Our FCM immunophenotype suggests that in addition to CD38 expression, the other markers such as CD7 are important to differentiate neoplastic basophils from reactive basophils.
Chromosomal abnormalities have been reported in CML patients with basophilia, [11],[12] acute basophilic leukemia [8] and other myeloid disorders with basophilia. [13] CBL with chromosomal aberrations has also been reported. [6] CBL is also shown to be associated with t(5;12)(q31;p13). [10] Interestingly, patients with the poorly defined entity, Ph-/BCR/ABL-atypical CML, have a significantly lower incidence of basophilia as compared with Ph+/BCR/ABL+ and Ph-/BCR/ABL+ CML. [3],[4] In our case, the PhͲ chromosome was detected with conventional GTG-banding. The fusion gene detection in malignancy is highly improved with dual color and dual fusion probes using FISH. In our case, the patient exhibited single fusion on chromosome 22 but fusion lacked on the der(9) chromosome. Since the signals are not found on any other chromosome, it was confirmed that the fusion on the der(9) chromosome was deleted. The deletions on der(9) have been reported in 15-20% CML patients. [14] Cytogenetic abnormalities have also been reported in acute basophilic leukemia. t(6;9)(p23;34) is reported to be associated with acute myeloid leukemia with basophils. [15] Chin et al. [16] extensively reviewed AML with t(6;9)(p23;q34) in basophilia cases and opined that t(6;9) results in a chimeric fusion gene between DEK (6p23) and CANNUP214 (9q34). In our case, the 9q34 region was deleted though the PhͲ chromosome was detected with GTG-banding. Hence, the deletion of 9q34 may be associated with basophilia in our case. Though various chromosomal abnormalities were involved in basophilic leukemia, the FISH with BCR/ABL dual fusion probe is important in CML cases to screen 9q34 deletion which may be associated with blast transformation.
In summary, CBL is a very rare form in myeloproliferative disorders and also seen in CML patients. However, the correlation of 9q deletion with basophilia has not been studied. The deletion of BCR/ABL fusion gene on the der(9) chromosome may be associated with basophilic leukemia and ultimately transformed to AML. Hence, the molecular studies on der(9q) may give some reasons to develop basophilia in CML patients.
 References References | |  |
| 1. | Denburg JA, Brownman G. Prognostic implications of basophil differentiation in chronic myeloid leukemia. Am J Hematol 1988;27:110-4. 
|
| 2. | Gill RM, Etzell JE. Basophilic leukemia in recurrent chronic myelogenous leukemia blast phase. Am J Hematol 2007;82:736-7. 
|
| 3. | Martiat P, Michaux JL, Rodhain J. Philadelphia negative (Ph-) chronic myeloid leukemia (CMl): Comparison with Ph+ CML and chronic myelomonocytic leukemia. The Groupe Francias de Cytogenetique Hematologique. Blood 1991;78:205-11. 
|
| 4. | Kurzrock R, Kantarjian HM, Shtalrid M, Gutternam JU, Talpaz M. Philadelphia chromosome negative chronic myelogenous leukemia without breakpoint cluster region rearrangement: A chronic myeloid leukemia with a distinct clinical course. Blood 1990;5:445-52. 
|
| 5. | Kyle RA, Pease GL. Basophilic leukemia. Arch Intern Med 1966;118:205-10. 
|
| 6. | Pardanani AD, Morice WG, Hoyer JD, Tefferi A. Chronic basophilic leukemia: A distinct clinico-pathologic entity. Eur J Haematol 2003;71:18-22. 
|
| 7. | Boniafazi F, De Vivo A, Rosti G, Tiribelli M, Russo D, Trabacchi E, et al. Testing sokal's and the new prognostic score for chronic myeloid leukemia treated with alpha-interferon. Italian co-operative study group on chronic myeloid leukemia. Br J Haematol 2000;111:587-95. 
|
| 8. | Peterson LC, Parkin JL, Arthur DC, Brunning RD. Acute basophilic leukemia: A clinical, morphologic and cytogenetic study of eight cases. Am J Clin Pathol 1991;96:160-70. 
|
| 9. | Han X, Jorgensen JL, Brahmandam A, Schlette E, Huh YO, Shi Y, et al. Immunophentypic study of basophils by multiparameter flow cytometry. Arch Pathol Lab Med 2008;132:813-9. 
|
| 10. | Tang G, Woods LJ, Wang SA, Brettler D, Andersen M, Miron PM, et al. Chronic basophilic leukemia: A rare form of chronic myeloproliferative neoplasm. Hum Pathol 2009;40:1194-9. 
|
| 11. | Hernández-Boluda JC, Cervantes F, Costa D, Carrió A, Montserrat E. Blast crisis of Ph-positive chronic myeloid leukemia with isochromosome 17q: Report of 12 cases and review of the literature. Leuk Lymphoma 2000;38:83-90. 
|
| 12. | Xue YQ, Guo Y, Lu DR, Gu J, Lu DW, Gong JX, et al. A case of basophilic leukemia bearing simultaneous translocations t(8;21) and t(9;22). Cancer Genet Cytogenet 1991;51:215-21. 
|
| 13. | Ma SK, Chan JC, Wan TS, Chan AY, Chan LC. Myelodysplastic syndrome with myelofibrosis and basophilia: Detection of trisomy 8 in basophils by fluorescence in-situ hybridization. Leuk Lymphoma 1998;31:429-32. 
|
| 14. | Sinclair PB, Nacheva EP, Leversha M, Telford N, Chang J, Reid A, et al. Large deletions at the t(9;22) breakpoints are common and may identify a poor prognosis subgroup of patient with chronic myeloid leukemia. Blood 2000;95:738-44. 
|
| 15. | Alsabeh R, Brynes RK, Slovak ML, Arber DA. Acute myeloid leukemia with t(6;9)(p23;q34): Association with myelodysplasia, basophilia and initial CD34 negative immunophenotype. Am J Clin Pathol 1997;107:430-7. 
|
| 16. | Chin Y, Lindgren V, Quigley S, Gaitonde S. Acute myelogenous leukemia with t(6;9)(p23;q34) and marrow basophilia: An overview. Arch Pathol Lab 2008;132:1835-7. 
|
[Figure 1], [Figure 2]
|