|
 
 |
| ORIGINAL ARTICLE |
|
| Year : 2014 | Volume
: 2
| Issue : 2 | Page : 50-55 |
|
Accelerated subclinical atherosclerosis in human immunodeficiency virus-infected patients on protease inhibitor regime
Gupta Salil1, Bhartiya Manish2, Nair Velu3, AK Tewari4, Naithani Nardeep5
1 Department of Internal Medicine, Armed Forces Medical College, Pune, India
2 Department of Medicine, Armed Forces Medical College, Pune, India
3 Department of Internal Medicine, Armed Forces Medical College, Pune/Office of the DGAFMS, New Delhi, India
4 Department Internal Medicine and Neurology, Armed Forces Medical College and Command Hospital, Pune/Commandant AFSMD, Mumbai, India
5 Department of Internal Medicine, Armed Forces Medical College, Pune/Commandant MH Wellington, Nilgiris, India
| Date of Acceptance | 13-Jun-2015 |
| Date of Web Publication | 3-Jul-2015 |
Correspondence Address:
Gupta Salil
Department of Internal Medicine, Armed Forces Medical College, Sholapur Road, Pune - 411 040, Maharashtra
India
 Source of Support: None, Conflict of Interest: None  | Check |
DOI: 10.4103/2321-9157.159969

Background: The life expectancy of human immunodeficiency virus (HIV) infected individuals has significantly increased following the introduction of combination antiretroviral therapy (cART). It is believed that because of their longevity these patients are more prone to atherosclerosis. This increased risk is not only linked to traditional risk factors for atherosclerosis, but also to HIV infection and cART. Objective: To identify the effects of HIV and cART (both protease inhibitor [PI] and non-PI based regimes) on carotid artery intima-media thickness (CIMT), a surrogate marker of subclinical atherosclerosis. Patients and Methods: Our cross-sectional study was done at a nodal HIV referral center. The study included 84 HIV-infected patients (24 exposed to PI-based cART, 30 to non-PI based cART, 30 not on cART) and 30 healthy age-matched controls. CIMT was measured ultrasonographically using high frequency B mode probe. The metabolic profile of the patients was also compared. Results: Using ANOVA the CIMT in patients on PI-based cART (0.82 + 0.17 mm) was significantly higher as compared to non-PI based cART (0.47 + 0.09 mm), HIV-infected patients not on ART (0.47 + 0.09 mm) and healthy control group (0.44 + 0.09 mm). When CIMT from non-PI based cART was compared with patients not on ART and healthy controls the difference was found not to be significant. Using linear regression analysis, the significant risk factors for this difference in CIMT in the PI-based regime included lower CD4 counts and higher total cholesterol. Conclusions: HIV-infected patients on PI-based cART are at risk for developing subclinical accelerated atherosclerosis. This will make them more prone to acute coronary syndromes, strokes, and peripheral vascular disease. Advanced HIV infection and high total cholesterol per se may also contribute to this risk of atherosclerosis. Screening using CIMT measurement and rigorous risk factor management is advocated for these patients.
Keywords: Accelerated atherosclerosis, carotid intima-media thickness, combination antiretroviral therapy, human immunodeficiency virus infection, protease inhibitor
How to cite this article:
Salil G, Manish B, Velu N, Tewari A K, Nardeep N. Accelerated subclinical atherosclerosis in human immunodeficiency virus-infected patients on protease inhibitor regime. J HIV Hum Reprod 2014;2:50-5 |
How to cite this URL:
Salil G, Manish B, Velu N, Tewari A K, Nardeep N. Accelerated subclinical atherosclerosis in human immunodeficiency virus-infected patients on protease inhibitor regime. J HIV Hum Reprod [serial online] 2014 [cited 2018 Feb 11];2:50-5. Available from: http://www.j-hhr.org/text.asp?2014/2/2/50/159969 |
| Introduction | |  |
Human immunodeficiency virus (HIV) infected patients are believed to be at increased risk for atherosclerosis and cardiovascular all-cause mortality including myocardial infarction and stroke. [1],[2] As the longevity of these patients is increasing the risk due to accelerated atherosclerosis is becoming significant. The factors responsible for this increased risk for atherosclerosis are not entirely clear. Some have shown this risk to be associated with chronic inflammation and endothelial dysfunction despite virological control with antiretroviral therapy. [3] Others have attributed it to traditional risk factors such as smoking, dyslipidemia, and advancing age. [4] There are also studies, which have reported increased risk with combination antiretroviral therapy (cART). [5]
The introduction of cART in clinical practice has resulted in a dramatic reduction of morbidity and mortality associated with HIV infection. However, several studies have demonstrated the development of cardiovascular risk factors, including dyslipidemia, insulin resistance, fat redistribution, and hypertension with use of the drugs, especially protease inhibitor (PI) based regimes. [6] Although PI-containing antiretroviral regimens have led to a notable extension of life expectancy in HIV-positive patients, prolonged lipid and glucose metabolism abnormalities due to these regimes could significantly affect the long-term prognosis and outcome of HIV-infected persons. An increasing concern is mounting particularly about the increased risk of acute coronary syndromes associated with new potent antiretroviral combinations. [7] In particular, some studies have demonstrated a correlation between antiretroviral therapy and increased risk of coronary heart disease.
The relationship between cerebrovascular disease and the use of cART in HIV-infected patients is still a matter of research. Several studies have investigated a possible association between antiretroviral treatment and cerebrovascular disease using various statistical approaches, but they often reported inconsistent and not comparable results. Classical vascular risk factors (male gender, age, family history of coronary heart diseases, smoking, arterial hypertension, diabetes, and dyslipidemia) obviously contribute to an increased risk of cerebrovascular complications in HIV-infected patients. On the other hand, recent data have highlighted systemic inflammation as a crucial factor in the pathogenesis of carotid lesions in HIV-positive subjects, [8] and Maggi et al. have evidenced that the ultrasonographic structure of the carotid lesions in HIV-infected individuals substantially differ from those of classical atherosclerotic plaques and share similar features with patients affected by arteritis. [9]
There is a lack of data from the developing world with respect to HIV, cART, and atherosclerosis. It is important to know the risk factors, which lead to the development of premature atherosclerosis in patients living with HIV infection so that appropriate management can be instituted in time. We designed this cross-sectional study with the aim of addressing the question that does HIV infection and the use of cART (both PI and non-PI based regimes) increase the chances of the patient developing atherosclerosis? We used carotid intima-media thickness (CIMT) as a surrogate marker of subclinical atherosclerosis. Measurement of CIMT by high-resolution ultrasonography is a well-accepted, noninvasive method of evaluating subclinical atherosclerosis, and is a potent predictor of myocardial infarction and stroke. [10]
| Materials and methods | |  |
This cross-sectional case-control study was performed at a tertiary care nodal reference center for HIV from October 2010 to July 2012. Patients with HIV infection, age between 18 and 45 years and who had the infection for at least 1-year from the time of detection were included in the study. All those on cART were included in the study only if they had been on the drugs for at least a period of 1-year. Patients were excluded if they had traditional risk factors for atherosclerosis specifically hypertension, diabetes, impaired glucose tolerance, past history of stroke, acute coronary syndrome or peripheral vascular disease. Age-matched controls were recruited from relatives of the patients who were HIV negative and did not have any of the atherosclerosis risk factors as mentioned earlier. Smoking, alcohol consumption, dyslipidemia, obesity, and family history of stroke or coronary artery disease were not considered as exclusion criteria either in the subjects or in the controls. The subjects were further subdivided into four groups as follows: Group 1 included patients on PI-based cART of at least 1-year duration, Group 2 included patients having HIV infection but not taking any form of antiretroviral treatment, Group 3 included those patients who were on non-PI based cART, and Group 4 included non-HIV infected individuals acting as control group. The study did not have any therapeutic implications; however clearance from the institutional ethical committee was obtained. Written informed consent was obtained from subjects as well as controls.
Detailed history was recorded from each patient about the duration of infection, details and duration of cART, past history of any cerebrovascular, cardiovascular or peripheral vascular disease, diabetes, hypertension, and smoking. All the subjects and controls underwent detailed clinical examination, which included recording of blood pressure, body mass index (BMI), and detailed physical examination. Laboratory tests included fasting plasma glucose, lipid profile, and CD4 count.
Visualization of CIMT was obtained via noninvasive high-resolution B-mode carotid artery ultrasonography and was performed by an experienced radiologist who was blinded to the HIV status and cART regimen of the patients. CIMT readings were taken from the carotid bulb or up to 1 cm of the proximal internal carotid artery. Average of six readings (three on each side) was taken. CIMT was expressed in millimeters.
Data was presented as mean ± standard deviation for descriptive data, while comparisons between groups were performed using ANOVA test (where appropriate), with significance levels placed at P < 0.05. Tuckey's P value test was used for pairwise comparison. Linear regression analysis was used to find out significant risk factors. SPSS software version 20 (IBM) was used for analysis.
| Results | |  |
There were 24 patients in Group 1 (HIV-infected patients on PI-based cART), 30 patients in Group 2 (HIV-infected patients not on any ART), 30 patients in Group 3 (HIV-infected patients on non-PI based cART), and 30 patients in the age-matched healthy controls. The CIMT was significantly increased in Group 1 (PI group) with P < 0.01 as compared to other groups using ANOVA. The P values for pairwise comparison using Tuckey's test for CIMT are given in [Table 1]. Group 1 (PI group) was significantly different from others; however there was no difference when Group 2 (HIV patients not on cART) and Group 3 (HIV patients on non-PI based cART) were compared to Group 4 (healthy controls) or to each other.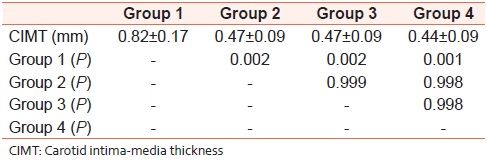 | Table 1: P values of comparison of individual groups with each other using ANOVA
Click here to view |
[Table 2] shows the values of various risk factors for atherosclerosis for each of the groups in the HIV setting. These include the traditional risk factors (which could not be excluded) and certain HIV-associated factors, which may influence results. Linear regression was used to test the relationship of these variables to CIMT (the dependent variable). The coefficients derived along with their significance are shown in [Table 3]. There was a significant relationship of CIMT with CD4 counts and total cholesterol with P < 0.05. These have also been shown in the form of a scatter plot [Figure 1] and [Figure 2]. CIMT was found to have a significant inverse relationship with CD4 count in that as the CD4 count fell the CIMT increased. Total cholesterol was also found to increase significantly with an increase in CIMT. Traditional risk factors such as age, sex, smoking, and BMI were not found to be significant among the groups. The duration of HIV infection and the duration of ART were also not found to be significant.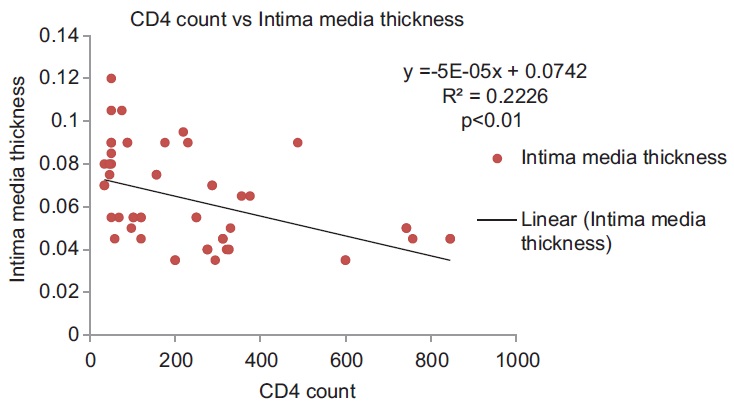 | Figure 1: Scatter plot showing significant inverse relationship of CD4 count with CIMT
Click here to view |
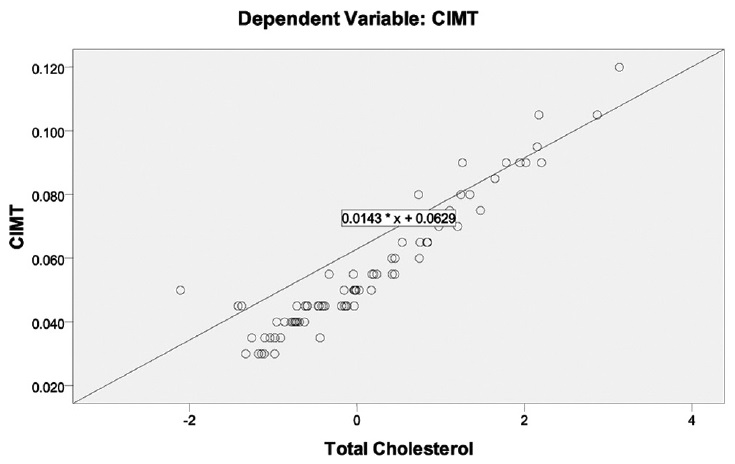 | Figure 2: Scatter plot showing significant direct relationship of coefficients of CIMT with total cholesterol
Click here to view |
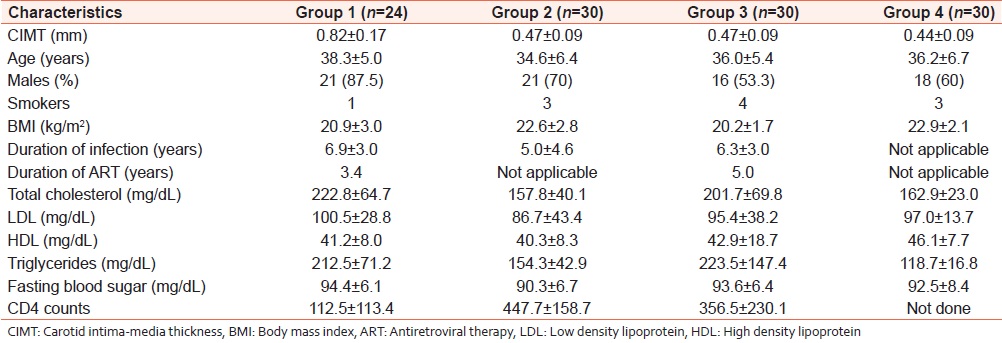 | Table 2: Values in each group of different atherosclerosis risk factors, which might influence CIMT as a marker of accelerated subclinical atherosclerosis
Click here to view |
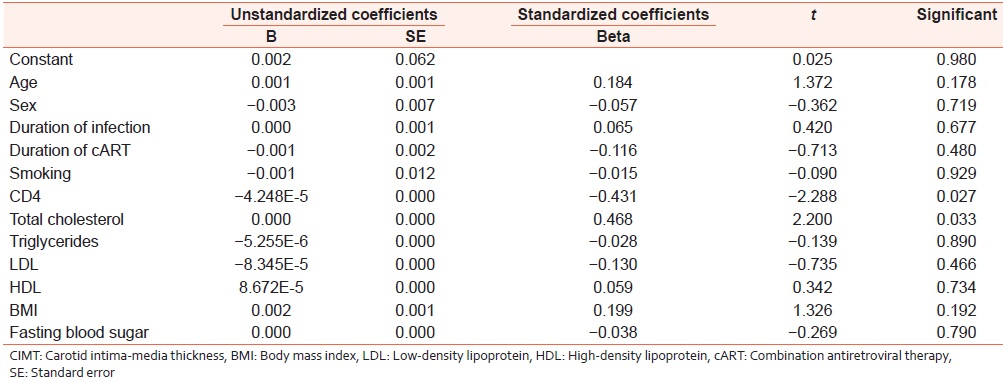 | Table 3: Coefficients and their P values of different risk factors obtained by linear regression analysis
Click here to view |
| Discussion | |  |
It has been long suspected that HIV-infected patients are more prone to develop accelerated atherosclerosis. Recent literature has several studies documenting the presence of accelerated atherosclerosis using different surrogate markers, both biochemical and radiological. However, the risk factors for the development of accelerated atherosclerosis in HIV patients are controversial. [11] Some have suspected HIV infection related endothelial dysfunction; [3] others have implicated therapy with ART, [4] while another group believes that the traditional risk factors have a role to play. [5],[12]
Our study reveals that CIMT (as a marker of accelerated subclinical atherosclerosis) is increased in a subset of HIV-infected patients on PI-based regime when compared to patients on non-PI based cART, patients not on ART and healthy age-matched controls. There was no significant difference in CIMT between HIV-infected patients on non-PI based cART, patients not on ART, and healthy age-matched controls. The other significant finding from our study is that CIMT has an inverse relationship with CD4 counts and direct relationship with total cholesterol. This may suggest that advanced HIV infection may also contribute to atherosclerosis. The significantly higher total cholesterol in Group 1 may be due to the PI effect or due to advance HIV infection.
Previous studies have shown a variable association between PI and CIMT in HIV-infected patients. A very recent study by Chanthong et al. showed that perinatally HIV-infected adolescents who were treated with PI-based therapy had higher CIMT as compared to non-PI based therapy. [13] Baker et al. showed that the use of PI therapy had a greater effect on the progression of CIMT in HIV-infected patients as compared to nonnucleoside inhibitor use. [14]
However, there are other studies, which have shown that there is no association between PI use and increase in CIMT. A meta-analysis done in 2009 by Hulten concluded that HIV infection and PI exposure were not strong independent risk factors of subclinical atherosclerosis. [15] CIMT was used as a marker of atherosclerosis. A study from Spain by Serrano-Villar et al. in 2012 did not find a significant association between nucleoside analog or PI use and increase in CIMT. [16] A more recent study by Rose et al. in 2013 concluded that there was no significant association between cART (both PI-based and non-PI based regimes) and progression of atherosclerosis. [17]
Our study adds strength to the evidence that PI-based regime in HIV-infected patients may contribute to accelerated atherosclerosis. The proposed mechanism by which PIs induce accelerated atherosclerosis is due to down-regulation of estrogen receptors. In a recent study by Xiang et al. ritonavir was shown to act as an antagonist for 17 β-estradiol in regulating α subtype estrogen receptor function and early events of atherosclerosis in Sprague-Dawley rats. [18]
The other significant finding from our study was that higher CIMT was associated with lower CD4 count. This finding may suggest a role of immunological changes in the pathogenesis of accelerated atherosclerosis in patients surviving with advanced HIV infection. Our study design does not permit that question to be answered, and it may be the topic for future research. It is also possible that the significant association of lower CD4 count in the subset of HIV patients on PI-based therapy and higher CIMT in our study may be biased. PI-based therapy is reserved for those who have failed first-line therapy. A patient with failed first-line therapy will invariably have falling or low CD4 counts and will be a candidate for PI-based therapy, and this may have contributed to the bias in that group. However, there are studies, which have shown that advanced HIV infection due to immune system dysregulation may itself be a risk factor for atherosclerosis supporting the finding in our study. [19],[20] A larger study designed to address that question is needed.
Dyslipidemia has been linked to the use of cART, especially PI-based therapy and the finding in our study may be linked to PI use. We also found that total cholesterol and triglyceride levels showed a higher but nonsignificant trend in the non-PI based cART group. This finding may merit further research with a larger number of patients.
Strength of our study is the presence of age-matched non-HIV-infected controls, group of patients not exposed to ART, and as far as possible excluding patients with traditional risk factors of atherosclerosis. The major limitation of this study is its cross-sectional design and relatively small number of patients as it is from a single center. There is no accepted absolute value of CIMT correlating to increased cardiovascular risk. The changes in CIMT when followed-up over a period of time correlate better with such risk. We measured CIMT and other metabolic parameters such as total cholesterol, low-density lipoprotein levels, and high-density lipoprotein levels at one particular time, thus the effect of chronic exposure to cART and these risk factors cannot be predicted unless these patients are followed-up over a period of time.
| Clinical implications | |  |
Patients living with HIV infection are prone to develop accelerated atherosclerosis. The pathogenesis of atherosclerosis is likely to be multi-factorial. Our study has demonstrated that patients on PI-based cART are more prone to develop subclinical atherosclerosis. Advanced HIV infection and dyslipidemia may also contribute to it. The accelerated atherosclerosis will make these patients more prone to acute coronary syndromes, myocardial infarction, strokes, and peripheral vascular disease. Hence, patients being placed on PI-based regimes and patients with advanced HIV infection must be strictly screened for traditional modifiable risk factors such as hypertension, diabetes, smoking, dyslipidemia, etc., and intervention planned accordingly. Alternative forms of cART may be considered if the patient has multiple preexisting atherosclerosis risk factors. These patients can be followed-up with periodic CIMT screening to document an increase from baseline.
| Directions for future research | |  |
Prospective follow-up studies with larger number of subjects can be planned to reinforce the findings from this study and to find out new risk factors, which make these patients more prone to atherosclerosis. Future direction of studies can include the role of statins.
| References | |  |
| 1. | Triant VA, Lee H, Hadigan C, Grinspoon SK. Increased acute myocardial infarction rates and cardiovascular risk factors among patients with human immunodeficiency virus disease. J Clin Endocrinol Metab 2007;92:2506-12.  |
| 2. | Obel N, Thomsen HF, Kronborg G, Larsen CS, Hildebrandt PR, Sørensen HT, et al. Ischemic heart disease in HIV-infected and HIV-uninfected individuals: A population-based cohort study. Clin Infect Dis 2007;44:1625-31.  |
| 3. | Pirš M, Jug B, Eržen B, Šabovic M, Karner P, Poljak M, et al. Relationship between markers of endothelial dysfunction and inflammation and subclinical atherosclerosis in HIV-infected male patients below 55 years of age. Acta Dermatovenerol Alp Pannonica Adriat 2014;23:49-52.  |
| 4. | Currier JS, Kendall MA, Zackin R, Henry WK, Alston-Smith B, Torriani FJ, et al. Carotid artery intima-media thickness and HIV infection: Traditional risk factors overshadow impact of protease inhibitor exposure. AIDS 2005;19:927-33.  |
| 5. | Barbaro G. Metabolic and cardiovascular complications of highly active antiretroviral therapy for HIV infection. Curr HIV Res 2006;4:79-85.  |
| 6. | Calza L, Manfredi R, Chiodo F. Dyslipidaemia associated with antiretroviral therapy in HIV-infected patients. J Antimicrob Chemother 2004;53:10-4.  |
| 7. | DAD Study Group, Friis-Møller N, Reiss P, Sabin CA, Weber R, Monforte Ad, et al. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med 2007;356:1723-35.  |
| 8. | Fisher SD, Miller TL, Lipshultz SE. Impact of HIV and highly active antiretroviral therapy on leukocyte adhesion molecules, arterial inflammation, dyslipidemia, and atherosclerosis. Atherosclerosis 2006;185:1-11.  |
| 9. | Maggi P, Perilli F, Lillo A, Carito V, Epifani G, Bellacosa C, et al. An ultrasound-based comparative study on carotid plaques in HIV-positive patients vs. atherosclerotic and arteritis patients: Atherosclerotic or inflammatory lesions? Coron Artery Dis 2007;18:23-9.  |
| 10. | Kablak-Ziembicka A, Tracz W, Przewlocki T, Pieniazek P, Sokolowski A, Konieczynska M. Association of increased carotid intima-media thickness with the extent of coronary artery disease. Heart 2004;90:1286-90.  |
| 11. | D'Abramo A, Zingaropoli MA, Oliva A, D'Agostino C, Al Moghazi S, De Luca G, et al. Immune activation, immunosenescence, and osteoprotegerin as markers of endothelial dysfunction in subclinical HIV-associated atherosclerosis. Mediators Inflamm 2014;2014:192594.  |
| 12. | Metkus TS Jr, Brown TT, Post WS. Cardiovascular disease associated with the human immunodeficiency virus: An update. Curr Treat Options Cardiovasc Med 2014;16:346.  |
| 13. | Chanthong P, Lapphra K, Saihongthong S, Sricharoenchai S, Wittawatmongkol O, Phongsamart W, et al. Echocardiography and carotid intima-media thickness among asymptomatic HIV-infected adolescents in Thailand. AIDS 2014;28:2071-9.  |
| 14. | Baker JV, Henry WK, Patel P, Bush TJ, Conley LJ, Mack WJ, et al. Progression of carotid intima-media thickness in a contemporary human immunodeficiency virus cohort. Clin Infect Dis 2011;53:826-35.  |
| 15. | Hulten E, Mitchell J, Scally J, Gibbs B, Villines TC. HIV positivity, protease inhibitor exposure and subclinical atherosclerosis: A systematic review and meta-analysis of observational studies. Heart 2009;95:1826-35.  |
| 16. | Serrano-Villar S, Estrada V, Gómez-Garre D, Ávila M, Fuentes-Ferrer M, Sánchez-Parra C, et al. Clinical factors and biomarkers associated with subclinical atherosclerosis in the human immunodeficiency virus infection. Med Clin (Barc) 2012;139:231-7.  |
| 17. | Rose H, Low H, Dewar E, Bukrinsky M, Hoy J, Dart A, et al. The effect of HIV infection on atherosclerosis and lipoprotein metabolism: A one year prospective study. Atherosclerosis 2013;229:206-11.  |
| 18. | Xiang J, Wang Y, Su K, Liu M, Hu PC, Ma T, et al. Ritonavir binds to and downregulates estrogen receptors: Molecular mechanism of promoting early atherosclerosis. Exp Cell Res 2014;327:318-30.  |
| 19. | McKibben RA, Margolick JB, Grinspoon S, Li X, Palella FJ Jr, Kingsley LA, et al. Elevated levels of monocyte activation markers are associated with subclinical atherosclerosis in men with and those without HIV infection. J Infect Dis 2015;211:1219-28.  |
| 20. | Karim R, Mack WJ, Kono N, Tien PC, Anastos K, Lazar J, et al. T-cell activation, both pre- and post-HAART levels, correlates with carotid artery stiffness over 6.5 years among HIV-infected women in the WIHS. J Acquir Immune Defic Syndr 2014;67:349-56.  |
[Figure 1], [Figure 2]
[Table 1], [Table 2], [Table 3]
|