|
 
 |
| ORIGINAL ARTICLE |
|
| Year : 2014 | Volume
: 2
| Issue : 1 | Page : 28-32 |
|
Residual monomer content of repair autopolymerizing resin after microwave postpolymerization treatment
Suraj Suvarna1, Tamanna Chhabra2, Deepti Raghav1, Devvrat Singh3, Prince Kumar1, Sukant Sahoo1
1 Department of Prosthodontics, Shree Bankey Bihari Dental College and Research Centre, Masuri, Ghaziabad, India
2 Department of Prosthodontics, K M Shah Dental College and Hospital, Vadodara, Gujarat, India
3 Department of Conservative Dentistry, Shree Bankey Bihari Dental College and Research Centre, Masuri, Ghaziabad, India
| Date of Web Publication | 12-Dec-2013 |
Correspondence Address:
Prince Kumar
Department of Prosthodontics, Shree Bankey Bihari Dental College and Research Centre, Masuri, Ghaziabad - 201 302, Uttar Pradesh
India
 Source of Support: None, Conflict of Interest: None  | Check |
DOI: 10.4103/2347-4610.122991

Purpose: Acrylic resins are one of the extensively used materials in removable prosthodontics however; complete dentures are highly vulnerable to fracture after long periods of clinical use. The repair of fractured prosthesis can be accomplished using acrylic resins that are light polymerized, autopolymerized, heat polymerized or microwave polymerized. Aim: To evaluate the residual monomer content of three different repair autoploymerising resins processed by two different polymerization techniques after microwave postpolymerization treatment. Materials and Methods: The study was executed with fabrication of a metal die and sixty test specimens of three different materials. The specimens were cured with conventional method and using pressure pot. After this the specimens were tested for residual monomer content using Fourier transform infrared spectroscopy. Results and Conclusion: Results confirmd that their residual monomer content decreased by subjecting them to another cycle of polymerization in microwave at 550 watts for 3 minutes. Keywords: Autopolymerizing resin, microwave, postpolymerization, residual monomer content
How to cite this article:
Suvarna S, Chhabra T, Raghav D, Singh D, Kumar P, Sahoo S. Residual monomer content of repair autopolymerizing resin after microwave postpolymerization treatment. Eur J Prosthodont 2014;2:28-32 |
How to cite this URL:
Suvarna S, Chhabra T, Raghav D, Singh D, Kumar P, Sahoo S. Residual monomer content of repair autopolymerizing resin after microwave postpolymerization treatment. Eur J Prosthodont [serial online] 2014 [cited 2018 Jul 5];2:28-32. Available from: http://www.eurjprosthodont.org/text.asp?2014/2/1/28/122991 |
| Introduction | |  |
Fracture at midline and repair of maxillary acrylic dentures is a common prosthodontic practice. [1],[2] When in function, midline fracture is the result of flexural fatigue failure caused by cyclic deformation of the base and is more likely to occur because flexure of the denture base occurs along the midline. [3] Most common causes of denture fracture are excessive bite force, improper occlusal plane, high frenal attachment, lack of balanced occlusion, and poor fit. [4],[5],[6] Denture repairs should have adequate strength, dimensional stability, [4],[7],[8],[9],[10],[11] good color match, easily, quickly performed, [12],[13],[14] and mechanical properties of the repair material can affect the strength of repaired denture. Heat-polymerized materials have been proven to have higher mechanical properties when compared with autopolymerized materials. [4],[6],[15] However, laboratory packing and flasking procedures are time-consuming and present risk of denture distortion by heat. [16] Therefore autopolymerizing repair resins are used for the same.
Degree of polymerization of autopolymerizing resin is less; hence, more residual monomer content is present which in turn affects its physical properties. [17] It can be further improved by following a period of immersion in hot water. [18],[19],[20] Free radicals continue to be present in the polymerized acrylic and it has been demonstrated that the residual monomer in an autopolymerizing acrylic resin may be reduced by further polymerization at these sites. [21] Microwave energy has also been a efficient source in curing acrylic resin. [22] However, the influence of microwave energy after polymerization has not been investigated. Therefore, the purpose of this investigation was to determine the amount of residual monomer content in autopolymerizing resin before and after microwave postpolymerization treatment.
| Materials and Methods | |  |
The study was completed by using three different autopolymerising resins classified as Group A (Acralyn'RRTM, Asian Acrylics, India), Group R (RRTM, Dentsply India PvtLtd.) and Group D (DPI-RR Cold CureTM, DPI, India), respectively [Figure 1]. Twenty specimens of each material were prepared. For making these specimens, a metallic die of dimension (64 × 12.5 × 3.5 mm) [Figure 2] was prepared. The metallic die was invested in denture flasks to prepare moulds. The mould was then packed with autopolymerizing resin mixed according to the manufacturer's instructions. Out of the 20 specimens of each group, 10 were allowed to cure in room temperature and pressure and the other ten were subjected to pressure pot curing for 20 min. The pressure pot was filled with water at temperature 40°C and internal pressure was regulated to 2.5 bars. The specimens were retrieved from flasks followed by finishing and polishing. Out of the 10 specimens prepared by two different curing methods of three different materials, five were kept as control and the other five were subjected to microwave postpolymerization treatment for 3 min at 550 watts [Table 1]. Specimens were then stored in water for 24 h at room temperature. | Figure 1: Test materials used in the study (from left to right acralyn RR, RR, and DPI-RR Cold Cure respectively)
Click here to view |
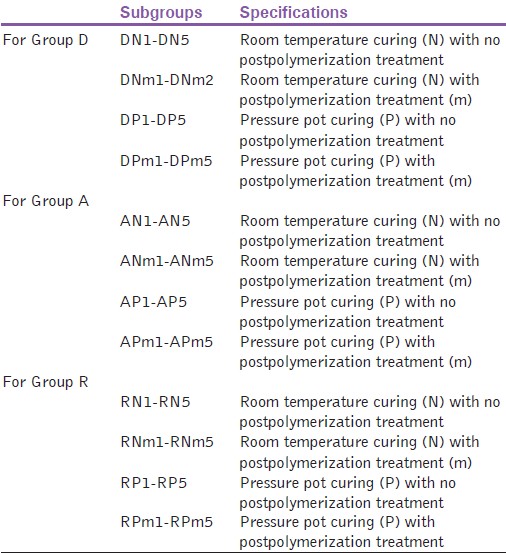 | Table 1: Sample nomenclature based on type of curing and microwave postpolymerization treatment for various groups
Click here to view |
The test was carried out using Fourier transform infrared spectroscopy (FTIR) Machine (Impact 400D-Nicolet Offenbach, Germany) [Figure 3]. FTIR technique has been used for qualitative and quantitative analysis of residual monomer during polymerization of polymethyl methacrylate. All the submitted samples were first cut into small pieces with the dimensions of 0.2 × 0.2 × 0.2 cm. The cut piece was then mounted on the FTIR (Fourier Transform Infrared) crystal [Figure 4]. It was observed that the monomer showed a very sharp peak at 1637 cm -1 due to substituted alkenes. C = C stretch from MMA (methyl methacrylate) appeared at 1637 cm -1 The peak at 1637 cm -1 was used for quantification of monomer present in polymer. | Figure 4: Cut piece mounted on the ATR/Fourier transform infrared spectroscopy crystal
Click here to view |
| Result and Conclusive Remarks | |  |
The residual monomer content was measured by FTIR machine (Impact 400D-Nicolet Offenbach, Germany) for all the 60 specimens. It was observed that the residual monomer content decreased for all the samples after microwave postpolymerization treatment [Table 2],[Table 3] and [Table 4]. There was statistically significant difference in the residual monomer content, before and after microwave postpolymerization treatment of all the three materials whether polymerized under normal conditions or in a pressure pot (P < 0.001) [Table 5]. The largest difference was seen in DPI material cured under room temperature and pressure.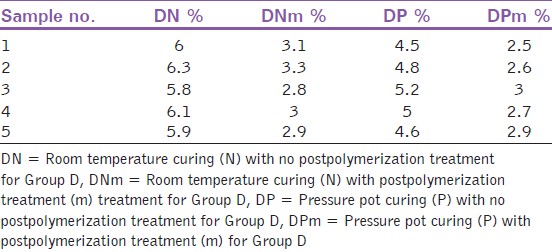 | Table 2: Residual monomer content for the samples of group D after microwave postpolymerization treatment
Click here to view |
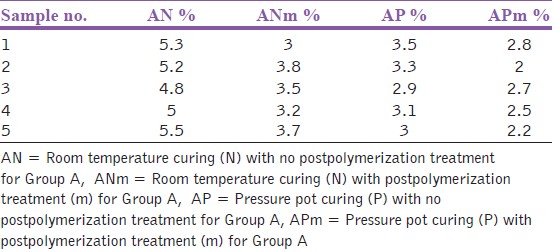 | Table 3: Residual monomer content for the samples of group A after microwave postpolymerization treatment
Click here to view |
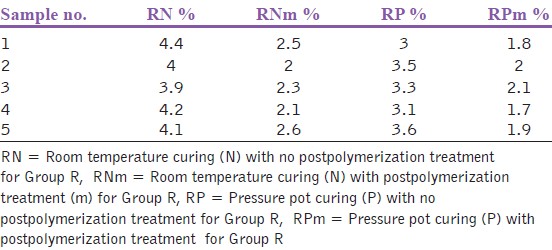 | Table 4: Residual monomer content for the samples of group R after microwave postpolymerization treatment
Click here to view |
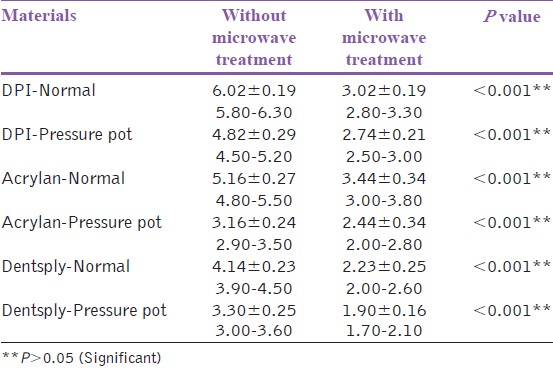 | Table 5: The residual monomer content, before and after microwave postpolymerization treatment with statistical interpretations
Click here to view |
| Discussion | |  |
Residual monomer content present in autopolymerized acrylic resins has restricted its usage as a choice of material for final prosthesis. Despite this, its properties like less time consumption for acralysation autopolymerizing resins are widely being used for repairing dentures. They are also being used in other areas like making denture bases, certain maxillofacial prosthesis, relining of dentures, provisional restoration, and so on. Hence, superior physical and mechanical properties are expected from autopolymerizing resins. Autopolymerizing resins by nature contain 3-5% of free monomer which affects the properties considerably. [23] This monomer if polymerized, improvement in the properties can be anticipated. Subjecting chemically cured resins to another short heat curing or pressure curing process was thus thought of. Subjecting these samples obtained by normal curing or pressure pot curing to microwave postpolymerization treatment could decrease the residual monomer content of the samples. It is in this context that the present study was carried out. Three different brands of repair autopolymerizing resin were selected to assess if there is any difference in the residual monomer content for various manufacturer. The samples obtained were subjected to an additional cycle of polymerization in a microwave oven at 550 W for 3 min. Residual monomer content of the samples was analyzed. In the present study, the materials were microwaved in dry conditions because uptake of water by the acrylic resin would lead to plasticization of acrylic resin, making it more flexible and resilient. Since microwave irradiation can be used to generate heat inside the resin, it may be that low monomer content achieved by the microwave treated specimens may be as a result of a higher degree of conversion of monomer into polymer. [24],[25] Largest effect of microwave was seen in DPI samples cured under normal conditions which was expected as it had the maximum amount of residual monomer content available to be cured further by microwave irradiation. Residual monomer, resulting from incomplete conversion of monomers into polymer, has the potential to cause irritation, inflammation, and an allergic response of the oral mucosa. Clinical signs and symptoms most frequently reported include erythematic, erosion of oral mucosa, and a burning sensation on the mucosa and tongue. [26]
Residual monomer concentration varies with the methods and the conditions of polymerization. [27] The variations in chemical composition and purity of the commercially available resin systems, the degree of conversion of their constituent monomers, and manipulative variables may all affect the biologic and physical properties of the acrylic resins. [28] Microwaves act only on the monomer, which decreases in the same proportion as the polymerization degree increases; therefore, the same amount of energy is absorbed by less and less monomer making the molecules increasingly active. [29] This is important because a self-regulation of the curing program takes place and leads to complete polymerization of the resin. In the microwave monomer molecules move by the high frequency electromagnetic field; their movements are the cause of internal heat generated. Some studies have shown that microwave-cured resins have decreased porosity because the heat necessary to break the benzoyl peroxide molecule into free radicals is created inside the resin once microwave radiations have greater penetrating capability. [30],[31] This way, the heat is dispersed more efficiently and polymerization is rapid and decreases residual monomer content as compared to warm water-bath-cured resin.
The results of the present investigation demonstrated that microwave irradiation is a method decreasing the residual monomer content of some denture base repair resins. This is in agreement with the studies conducted earlier. [24],[27],[28],[30] Thus, the present study supports the statement that another short cycle of microwave irradiation can reduce the residual monomer content of autopolymerizing resins. Because of s, the power/time combinations were found appropriate for the material; therefore, it can be expected that this reduction may have a contributory effect upon mechanical properties of these materials as well as decrease in the possibility of cytotoxicity and tissue reactions. Though, the limitations of the study were as follows:
- Masticatory load applies to the denture repair material and to the denture base as a whole, rather than to the repair material alone. The present study did not simulate the intraoral environment to evaluate the mechanical response of the samples and properties at the repair resin/denture base resin interface
- Only one power and time combination was used. Any variation in the both and its effect on residual monomer were not tested.
| References | |  |
| 1. | Beyli MS, von Fraunhofer JA. An analysis of causes of fracture of acrylic resin dentures. J Prosthet Dent 1981;46:238-41. 
[PUBMED] |
| 2. | Darbar UR, Huggett R, Harrison A. Denture fracture-a survey. Br Dent J 1994;176:342-5. 
[PUBMED] |
| 3. | Shen C, Colaizzi FA, Birns B. Strength of denture repairs as influenced by surface treatment. J Prosthet Dent 1984;52:844-8. 
[PUBMED] |
| 4. | Beyli MS, von Fraunhofer JA. Repair of fractured acrylic resin. J Prosthet Dent 1980;44:497-503. 
[PUBMED] |
| 5. | Polyzois GL, Andreopoulos AG, Lagouvardos PE. Acrylic resin denture repair with adhesive resin and metal wires: Effects on strength parameters. J Prosthet Dent 1996;75:381-7. 
|
| 6. | Agarwal M, Nayak A, Hallikerimath RB. A study to evaluate the transverse strength of repaired acrylic denture resins with conventional heat-cured, autopolymerizing and microwave-cured resins: An in vitro study. J Ind Prosthodont Soc 2008;8:36-41. 
|
| 7. | Stipho HD, Stipho AS. Effectiveness and durability of repaired acrylic resin joints. J Prosthet Dent 1987;58:249-53. 
|
| 8. | Ward JE, Moon PC, Levine RA, Behrendt CL. Effect of repair surface design, repair material, and processing method on the transverse strength of repaired acrylic denture resin. J Prosthet Dent 1992;67:815-20. 
|
| 9. | John J, Gangadhar SA, Shah I. Flexural strength of heat-polymerized polymethyl methacrylate denture resin reinforced with glass, aramid, or nylon fibers. J Prosthet Dent 2001;86:424-7. 
|
| 10. | Ng ET, Tan LH, Chew BS, Thean HP. Shear bond strength of microwaveable acrylic resin for denture repair. J Oral Rehabil 2004;31:798-802. 
|
| 11. | Polyzois GL, Tarantili PA, Frangou MJ, Andreopoulos AG. Fracture force, deflection at fracture, and toughness of repaired denture resin subjected to microwave polymerization or reinforced with wire or glass fiber. J Prosthet Dent 2001;86:613-9. 
|
| 12. | Dar-Odeh NS, Harrison A, Abu-Hammad O. An evaluation of self-cured and visible light-cured denture base materials when used as a denture base repair material. J Oral Rehabil 1997;24:755-60. 
|
| 13. | Polyzois GL, Handley RW, Stafford GD. Repair strength of denture base resins using various methods. Eur J Prosthodont Restor Dent 1995;3:183-6. 
|
| 14. | Harrison WM, Stansbury BE. The effect of joint surface contours on the transverse strength of repaired acrylic resin. J Prosthet Dent 1970;23:464-72. 
|
| 15. | Anusavice KJ. Philips science of dental materials. 11 th ed. Bangalore: Prism Saunders, Prism book Pvt Ltd; 2002, p. 124-36, India 
|
| 16. | Murphy WM, Huggett R, Handley RW, Brooks SC. Rigid cold curing resins for direct use in the oral cavity. Br Dent J 1986;160:391-4. 
|
| 17. | Lamb DJ, Ellis B, Priestley D. The effects of process variables on levels of residual monomer in auto-polymerising dental acrylic resin. J Dent 1983;11:80-8. 
|
| 18. | Azzarri MJ, Cortizo MS, Alessandrini JL. Effect of the curing conditions on the properties of an acrylic denture base resin microwave-polymerised. J Dent 2003;31:463-8. 
|
| 19. | Ilbay SG, Güvener S, Alkumru HN. Processing dentures using a microwave technique. J Oral Rehabil 1994;21:103-9. 
|
| 20. | De Clerck JP. Microwave polymerization of acrylic resins used in dental prosthesis. J Prosthet Dent 1987;57:650-8. 
|
| 21. | Vergani CE, Seo RS, Pavarina AC, dos Santos Nunes Reis JM. Flexural strength of auto-polymerizing denture reline resins with microwave post-polymerization treatment. J Prosthet Dent 2005;93:577-83. 
|
| 22. | Weaver RE, Goebel WM. Reactions to acrylic resin dental prosthesis. J Prosthet Dent 1980;43:138-42. 
|
| 23. | Kedjarune U, Charoenworaluk N, Koontongkaew S. Release of methyl methacrylate from heat cured and auto-polymerized resins: Cytotoxicity testing related to residual monomer. Aust Dent J 1999;44:25-30. 
|
| 24. | Kalipçilar B, Karaaðaçlioðlu L, Hasanreisoðlu U. Evaluation of the level of residual monomer in acrylic denture base materials having different polymerization. J Oral Rehabil 1991;18:399-401. 
|
| 25. | Sadamori S, Ganefiyanti T, Hamada T, Arima T. Influence of thickness and location on the residual monomer content of denture base cured by three processing methods. J Prosthet Dent 1999;72:19-22. 
|
| 26. | Jacob J, Chia LH, Boey FY. Microwave polymerization of poly (methymetacrylate): Conversion studies at variable power. J Appl Polym Sci 1997;63:787-9. 
|
| 27. | Shim JS, Watts DC. Residual monomer concentrations in denture base acrylicresin after an additional, soft-liner, heat cure cycle. Dent Mater 1999;15:296-300. 
|
| 28. | International standards organization ISO 1567 for denture base polymers. Geneva: ISO; 1998. 
|
| 29. | Doðan A, Bek B, Cevik NN, Usanmaz A. The effect of preparation conditions of acrylic denture base materials on the level of residual monomer, mechanical properties and water absorption. J Dent 1995;23:313-8. 
|
| 30. | Rached RN, Del-Bel Cury AA. Heat-cured acrylic resin repaired with microwave-cured one: Bond strength and surface texture. J Oral Rehabil 2001;28:370-5. 
|
| 31. | Patil PS, Chowdhary R, Mandokar RB. Effect of microwave post-polymerization treatment on residual monomer content and the flexural strength of auto-polymerizing reline resin. Indian J Dent Res 2009;20:293-7. 
[PUBMED]  |
[Figure 1], [Figure 2], [Figure 3], [Figure 4]
[Table 1], [Table 2], [Table 3], [Table 4], [Table 5]
|