Paradoxical Role of T-type Calcium Channels in Coronary Smooth Muscle
Calcium influx via voltage-gated channels plays important roles in muscle excitation-contraction coupling, hormone and neurotransmitter stimulus-secretion coupling, regulation of gene transcription, and neuronal firing. To fulfill these diverse roles, mammals have evolved ten genes encoding voltage-gated calcium channels (Cav channels) (Figure 1⇓). These channels also serve as important drug targets, with most therapeutically useful calcium-channel blockers targeting L-type channels, such as Cav 1.2, found on cardiac or smooth muscle. These drugs (e.g., amlodipine) are useful in the treatment of high blood pressure because they block Ca2+ influx via L-type channels into smooth muscle cells (SMC), which results in relaxation.
SMCs also express T-type calcium currents (1). The development and characterization of mibefradil (also termed Ro 40-5967), a novel antihypertensive drug with modest selectivity for T-type channels (2), supported the hypothesis that blocking T-type channels would produce vasorelaxation. Mibefradil was shown to be an effective antihypertensive drug that produced coronary and peripheral vasodilation without triggering baroreceptor reflexes (2). Although mibefradil was withdrawn from clinical use, it continues to be a useful tool in the research laboratory. For example, studies on mibefradil’s actions have implicated a role for T-type channels in pain perception (3–5). However, other studies have questioned the selectivity of mibefradil, showing that the drug can also block other voltage-gated calcium channels (6), sodium channels (7), potassium channels (8–10), non-selective cation currents (11), and even volume-activated chloride currents (12). These results have cast doubt on the hypothesis that mibefradil’s antihypertensive action is because of blockage of T-type channels.
In order to elucidate the physiological roles of T-type channels, Chen et al. generated mice lacking Cacna1H (one of the three known T-type channel genes), which encodes the Cav 3.2 or α1H channel (13). In contrast to the other two T-type channel genes, which are predominantly expressed in brain, Cacna1H is more broadly expressed in peripheral tissues, including heart, liver, and kidney (14). Cav 3.2 is also expressed in adrenal cortex (15), where it participates in the secretion of aldosterone and cortisol (16), and in embryonic skeletal muscle, where it plays a key role in myoblast fusion (17). Additionally, Cav 3.2 mRNA is abundantly expressed in dorsal root ganglion (DRG) neurons (18). Of historical note, low voltage–activated T-type currents were originally isolated from high voltage–activated currents by voltage-clamp analysis of DRG neurons (19–21). Subsets of DRG neurons express extremely large T-type currents, suggesting that these currents are important for sensory processing. However, the role of T-type currents on sensory processing might be quite different than that of regulating intracellular calcium concentrations, and may reflect the originally ascribed role of T-type currents as a pacemaker, capable of generating low-threshold spikes that in turn initiate Na+ -dependent action potentials (22). To show that Cav 3.2 is necessary for the formation of T-type currents, Chen and coworkers isolated neonatal DRG neurons from Cav 3.2−/− mice and recorded their calcium currents using voltage-clamp techniques. These studies showed the complete ablation of T-type current with no compensation. If T-type channels were a target for novel analgesic drugs, then one would predict that these mice would show diminished responses to painful stimuli. Unfortunately, this hypothesis was not tested. Instead, Chen and coworkers used the Cav 3.2−/− mice to study coronary artery contraction (13). Their surprising conclusion was that rather than mediating contraction, T-type currents mediate relaxation of coronary smooth muscle.
In contrast to littermate controls, Cav 3.2−/− mice were smaller, had malformations in the spinal column, trachea, and coronary arteries, and developed cardiac fibrosis (13, 23). Surprisingly, following preconstriction with the thromboxane analog U46619, treatment with acetylcholine caused further constriction of isolated coronary arteries from Cav 3.2−/− mice rather than the dilation observed in similarly treated control arteries. In addition, coronary arteries from Cav 3.2−/− mice were less sensitive to the vasodilatory effects of the nitric oxide donor, sodium nitroprusside. The connection between T-type channels and nitric oxide mediated relaxation remains unclear. Perhaps gene array experiments would be useful to identify whether the expressions of other components of this pathway are affected.
Chen and coworkers focused on the possible association of Cav 3.2 and calcium-activated potassium channels (BKCa), showing that both channels sediment to the bottom of a sucrose gradient, and that antibodies directed against Cav 3.2 are capable of precipitating a protein recognized by antibodies directed against BKCa (13). If Ca2+ influxes via T-type channels are an important regulator of BKCa channels, then there may be less activation of BKCa channels in SMCs from Cav 3.2−/− mice. However, similar responses were observed in normal and in Cav 3.2−/− mice to the BKCa agonist NS-1619 (13). Although neuronal T-type currents have been implicated in the activation of calcium-activated K+ channels, these channels were voltage-independent SKCa channels (24). In contrast, neuronal BKCa channels are thought to be activated by Ca2+ influx via high voltage–activated Ca2+ channels (25). Clearly, more work is needed to establish a functional role for the association of Cav 3.2 and BKCa channels.
Studies on transgenic Cav 3.2−/− mice have provided definitive evidence that T-type channels produce low threshold Ca2+ spikes that control burst firing of thalamic neurons, and have provided insights into the role of burst firing in sensory gating (5, 26). Perhaps future studies on Cav 3.2−/− mice will provide meaningful insights into the role of these channels in pain, adrenal hormone secretion, and neuronal signaling.
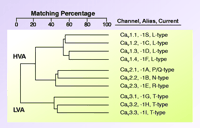
Family of voltage-gated calcium channels. Cloning of mammalian Ca2+ channels revealed the existence of ten genes. Alignment of their deduced amino acid sequences indicates that these channels can be grouped into three subfamilies, providing the basis for a rational nomenclature scheme. This family tree is based on the full-length human sequences, and differs from that obtained using membrane spanning regions (1). The Cav 1 channels carry L-type currents, and play critical roles in muscle excitation-contraction coupling. They are also the target of traditional “calcium channel blockers.” The Cav 2 channels are a second subfamily of high voltage–activated channels (HVA), and they play a dominant role in neurotransmitter release. Although subtype-selective small-molecule drugs have not been developed to for Cav 2 channels, these channels can be distinguished with peptide toxins. The Cav 3 channels carry T-type currents. These channels open after small depolarizations of the plasma membrane, and hence are also termed low voltage–activated (LVA) channels. This property allows Cav 3 channels to act as a pacemaker current, such as that originally observed in low threshold spiking and rebound burst firing of neurons (22).
- © American Society for Pharmacology and Experimental Theraputics 2004
References

Edward Perez-Reyes, PhD, is an Associate Professor in the Department of Pharmacology at the University of Virginia. He is supported by NIH Grant NS038691. Email: eperez{at}virginia.edu.