REGENERATIVE PHARMACOLOGY: THE FUTURE IS NOW
Abstract
Regenerative medicine technologies cross the boundaries of numerous scientific fields, including cell and molecular biology, chemical and material sciences (i.e., nanotechnology), engineering, molecular genetics, physiology and pharmacology. As such, regenerative medicine truly represents an integrative and logical (r)evolution of medical science. This groundbreaking field of research has the potential to radically alter the treatment of diseases or disorders characterized by the lack of viable cells or tissues. The goal of this report is to review the current challenges and opportunities in the emerging field of regenerative medicine and to describe the role of the pharmacological sciences in the acceleration, optimization, and evaluation of engineered tissue function in the service of regenerative medicine technologies.
Introduction
Regenerative medicine represents a global, groundbreaking, and interdisciplinary biotechnological effort with tremendous potential to promote and extend the quality of life of the aging US and world population (1). Tissue engineering, stem cell therapy, and cloning technologies are subsumed under the rubric of regenerative medicine. Tissue engineering is one of the major components, which, in turn, combines the fields of cell transplantation, materials science, and engineering to acquire medicinal tissues that could restore and maintain normal tissue/organ function, including endogenous regenerative capacity (2). The widespread promise of regenerative medicine has been recently codified in government publications (1).
From an economic point of view, regenerative medicine is positioned to play a major role in US and worldwide healthcare, especially in consideration of the current $350 billion–world market for replacement organ therapies. The US market for regenerative medicine, in the range of $100 billion, would have a significant impact on job growth in the healthcare industry. “The worldwide market for regenerative medicine is conservatively estimated to be $500 billion by 2010” (3). From this standpoint, regenerative medicine technologies could be an important step to abate the impending health care cost explosion that will be ushered in by an aging population, although the decades of research conducted thus far have been witness to limited clinical success (1, 2, 4–6). Nevertheless, the field has made great strides in recent years and it seems certain that major advances are on the horizon. In fact, a search of the Medline database using the term ”tissue engineering” revealed 1701 review articles, with approximately 80% of these appearing in the last five years. Clearly, this is a burgeoning field of research, ripe for major medical breakthroughs.
A detailed recitation of the challenges and opportunities encompassing all aspects of regenerative medicine technologies is well beyond the scope of this report, and the reader is referred elsewhere for reviews on the subject (7, 8, 50). The explicit goal of this review is to outline the importance of applied pharmacology to regenerative medicine and the tissue engineering process. As a necessary prerequisite to discussion of the role of integrative and applied pharmacology to regenerative medicine/tissue engineering, we first review some of the basic steps in the tissue engineering process.
Construction of a Tissue or Organ
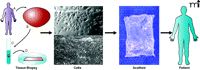
The construction of bioengineered tissues is a multi-step process (2, 5). As illustrated in Figure 1⇓, the first step is the identification and harvest of somatic cells, for which autologous (and thus immunocompatible) cells derived from the tissue of interest would be the preferred choice. However, there will inevitably be instances in which biopsies are not possible or in which tissues are so severely damaged as to render the isolation and expansion of autologous cells from those tissues impossible. In such a scenario, stem cells, derived from embryonic, amniotic, cord blood, placental, or bone marrow sources, become attractive resources (2, 5, 9).
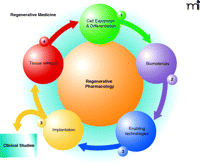
The central role of pharmacology in regenerative medicine and tissue engineering.Regenerative pharmacology would provide both a screening and optimization tool for accelerated maturation and function of bioengineered tissues. 1. Cell expansion and Differentiation signifies cell expansion and differentiation via modulation/manipulation of growth factors, cytokines, hormones, neurotransmitters and neuromodulators-both before and after scaffold seeding. 2. Biomaterials involve the use of biodegradable, biocompatible, cell and tissue-specific scaffolds to modulate cellular phenotype/pharmacology and promote cellular integration and tissue formation in vitro and in vivo. 3. Enabling Technologies will use nanotechnology in the development of “smart” scaffolds for sustained or burst release of drugs, hormones, chemicals, growth factors. Bioreactors are center-stage for tissue preconditioning and optimization of cellular phenotype, tissue organization, maturation and function, as is the development of non-invasive pharmacological evaluation of engineered tissues. Thus, it is clear that enabling technologies will be required throughout the tissue engineering process. However, we illustrate its contribution at this point in the process to emphasize the importance of this particular step to mainstream pharmacology (see Table 1). 4. Implantation includes the functional evaluation of engineered tissues. Again, drug and cell delivery technologies can be utilized to further accelerate and enhance tissue maturation, integration, and function. VEGF, NGF, for example, may be provided via encapsulation techniques to promote accelerated in growth of vessels and nerves, respectively, into the engineered tissue. 5. Tissue biopsy involves the pharmacological characterization of cell phenotype and tissue function. Again, during the preclinical development phase of the tissue engineering process, one would assess membrane receptors, signal transduction, excitation-contraction coupling. This information is then used to evaluate the existing protocol and thereby direct optimization of the tissue engineering process.
The next steps in the process involve cell growth and expansion. The goal is to derive sufficient numbers of cells with the appropriate phenotypic and functional characteristics that can be “seeded” onto a matrix system, or scaffold. The cell-seeded scaffolds are then either directly implanted into the host, or they may be further incubated under physiologic conditions; in the latter case, cell-seeded scaffolds can be preconditioned in “bioreactors” (see below). Regardless of the precise incubation conditions, the concept remains the same—once the bioengineered constructs have been appropriately acclimated, they are implanted into the host.
What Is Regenerative Pharmacology?
What is the role of pharmacology in regenerative medicine? Regenerative pharmacology, for the purposes of this report, can be broadly defined as the application of pharmacological sciences to accelerate and optimize (either in vitro or in vivo) the maturation and function of bioengineered tissues. As such, pharmacology should assume a critical role in all aspects of the regenerative process (Figure 2⇓). Serving a dual role, pharmacological methods can facilitate the development of the enabling technologies required for accelerated cellular and tissue maturation and function, as well as to characterize bioengineered tissue function to ensure their reliability and utility. Some examples of this are provided in Figure 2⇓.
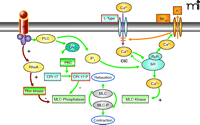
To further emphasize this point, we will briefly review recent developments in tissue-engineered urinary neobladder and large-caliber tissue-engineered blood vessels. These two examples illustrate how pharmacological tools and methods can be iteratively applied in tissue engineering. The focus here is on bioengineered constructs in which the smooth muscle cell is a primary parenchymal cell type, and thus, smooth muscle cell tone, that is, contraction and relaxation, is of major importance. A putative template for native myocyte pharmacology and excitation-contraction coupling is shown in Figure 3⇓. The principles outlined here will have broad applications for regenerative medicine that may involve other cell types and engineered tissues.
Major signal transduction pathways that modulate myocyte tone. Aspects of these pathways could serve as a benchmark for phenotypic characterization of bioengineered muscle. [(+) denotes a positive or stimulatory effect; (−) denotes a negative or inhibitory effect. BKCa, the calcium-sensitive K channel (maxi-K), although clearly there are several types of K channels known to be present in detrusor myocytes; SR, sarcoplasmic reticulum; RyR, ryanodine receptor; PLC, phospholiapse C; DAG, diacylglycerol; PKC, protein kinase C; IP3, inositol trisphosphate; MLC, myosin light chain; A, agonist; R, receptor; CPI-17, protein kinase C-potentiated protein phosphatase-1 inhibitory protein.]
Development of an Engineered Bladder
The recent report that tissue-engineered neobladders had been successfully implanted in patients, with up to 5 years of follow-up, generated much excitement about the potential of regenerative medicine (10). These studies followed the process outlined in Figure 1⇑ and represented the culmination of more than a decade of preclinical work. A more detailed description of this achievement can be found in the section below on clinical applications of regenerative medicine; here, the focus is on regenerative pharmacology and smooth muscle.
Cell Source
Smooth muscle cells are well known for losing some of their phenotypic characteristics when placed into cell culture environments (11, 12, 51, 52), and bladder smooth muscle cells are no exception. When maintained in two-dimensional cultures, bladder myocytes dedifferentiate and lose their ability to contract in response to appropriate agonists (13). Moreover, it is not known to what extent these dedifferentiated cells are able to redevelop after implantation into the host (14), which is a major concern, given that cellular phenotype will determine proper functioning of engineered bladder tissue after implantation.
In this regard, Lai and coworkers (13) studied human smooth muscle cells derived from functionally normal or dysfunctional bladders (i.e., exstrophic bladders and neurogenic bladders). These researchers cultivated, expanded, and seeded bladder cells onto biodegradable polymer scaffolds, which were then implanted into athymic mice. After two months, the cell-seeded scaffolds demonstrated the typical contraction–relaxation response to supramaximal electrical field and carbachol stimulation. The authors concluded that tissue-engineered muscles from normal and diseased bladders retain important aspects of their phenotype in vitro and as well as after implantation, and that there were no phenotypic or functional differences between muscle cells obtained from urodynamically normal or pathological bladders. In short, these observations suggest that bladder muscle cells, regardless of their origin, may have the potential to be engineered into functional bladder tissues.
Biomaterials and Scaffolds
The development and application of biomaterials are essential to tissue engineering in regenerative medicine (15). Scaffolds provide mechanical support and shape for the developing bioengineered tissue in vitro and post implantation, allowing seeded cells to expand, differentiate, and organize. Generally, three main types of biomaterials have been used for tissue engineering: 1) naturally derived materials (e.g., fibrin, collagen, chitosan, and alginate); 2) synthetic polymers (e.g., polyglycolic acid [PGA], polylactic acid [PLA], polylactic-coglycolic acid, [PLGA], polyhydroxyalkanoate [PHA], and polydioxanone [PDS]; and 3) acellular tissue matrices (e.g., bladder lamina propria and small intestinal submucosa). In the engineering of neobladders, synthetic polymers have been utilized as scaffolds in which the macrostructure, mechanical properties, and degradation can be readily controlled and manipulated. Thus, Oberpenning and colleagues (16), who reconstituted the first functional dog urinary bladder, used PGA matrix configured into a bladder-shaped mold, which was coated with PLGA in order to achieve adequate mechanical strength. A biodegradable composite scaffold made of collagen and PGA/PLGA was also used for implantation of bladder constructs in humans by Atala and coworkers (10). The true importance of such biodegradable scaffolds is that their degradation can prevent a chronic foreign body response.
Implantation of the Bladder Construct
After configuration of the bladder mold, the scaffold is typically seeded with urothelial cells on the inside of the scaffold and smooth muscle cells on the outside of the scaffold; it is then placed in an incubator for a period of three to four days. At that point, the bladder construct can be implanted in the host (i.e., dog or human). In functional evaluations for up to eleven months after implantation in the dog, the neobladders demonstrate a normal capacity to retain urine, normal elastic properties, and normal histological architecture (16). In humans, biopsies of engineered bladders show an adequate structural architecture and phenotype, which correlate with improvements in bladder volume and compliance (10).
Development of Tissue-Engineered Blood Vessels
The development of tissue-engineered vasculature could have an enormous impact in biomedicine and has consequently received widespread attention (17–24). Small- as well as large-diameter vessels must be considered in the construction of vascular grafts, and a variety of synthetic and naturally derived biomaterials have been employed thus far. The goal is always the same, that is, to develop a reproducible, biocompatible scaffold that recapitulates the native vasculature.
Cell Source
As with the bioengineered bladder, a variety of cell sources are available for tissue-engineered blood vessels. In addition to autologous explant cultures of somatic cells, circulating progenitor cells (both endothelial and smooth muscle progenitors) may also be utilized (26, 30). The phenotypic diversity of vascular smooth muscle cells is well documented (11, 12, 25), undoubtedly reflecting the variety of environmental and endogenous developmental cues in the vessel wall to which vascular smooth muscle cells respond (24). Biochemical, mechanical, and extracellular matrix signals are important parameters to smooth muscle cell function and must be considered in the engineering of blood vessels.
Biomaterials
From a biological perspective, decellularized vessels (e.g., excised vessels devoid of cellular material) retain the biochemical composition, ultrastructural architecture, and biomechanics of native vessels. Tissue-engineered blood vessels have been designed using decellularized collagen-based vascular scaffolds derived from porcine blood vessels (26, 27). Synthetic scaffolds nevertheless offer off-the-shelf customizable advantages in the clinical setting.
Construction of Tissue-Engineered Blood Vessels
Many different approaches have been evaluated for the construction of tissue-engineered blood vessels (17–24, 26). In fact, a recent review of this subject identified more than three dozen reports of tissue-engineered blood vessels since the early reports of Weinberg and Badylak (28, 29), half of which involved engineered vessels that were subsequently implanted in vivo (30). A few examples will serve to make the point.
L’Heureux and colleagues (31) were among the first to develop a tissue engineering approach for the production of completely biological blood vessels from cultured human cells. These tissue-engineered blood vessels were designed to be vascular grafts and contained the three cell types found in natural vessels: endothelial cells, vascular smooth muscle cells, and fibroblasts. To obtain these engineered vessels, human vascular smooth muscle cells from umbilical cords were cultured; after fifteen to twenty days of culture, cells formed thick sheets comprised of cells and extracellular matrix that could be peeled off from the culture flask. These sheets were wrapped around a styrene tubular support to produce a cylinder composed of approximately five concentric sheet layers. After a week of maturation, the layers adhered to one another, thereby forming a cohesive tubular tissue. The tissue, left remaining on the tubular support, was cut into rings, which were maintained in culture for pharmacological analysis. Specifically, contractile activity was recorded isometrically with a force transducer in response to vasomodulators such as histamine, bradykinin, ATP, and UTP (32). The investigators further demonstrated that sustained agonist-induced contractions were associated with transient increases in cytosolic Ca2+ concentration, suggesting sensitization of the contractile machinery to Ca2+.
Other investigators have used suspensions of cultured smooth muscle cells from the medial layer of bovine aorta, seeded onto PGA scaffolds (22). After eight weeks of culture in a bioreactor under pulsatile conditions, vessels were obtained that manifested a gross appearance identical to that of native arteries. However, the tension induced by prostaglandin F2α in these engineered vessels was only 5% of that of native controls.
Decellularized collagen-based vascular scaffolds derived from porcine blood vessels have also been successfully used for tissue-engineered blood vessels in vivo (26). Kaushal and colleagues used endothelial progenitor cells from peripheral blood of lambs. After expansion these cells were seeded on decellularized iliac vessels. The grafts were used for carotid interposition in sheep for 130 days and remained patent throughout the duration. Retrieved grafts exhibited contraction in response to noradrenaline and serotonin and relaxation after exposure to acetylcholine. Further, the observed relaxation responses were attenuated by the nitric oxide synthase inhibitor, l-NAME, indicating that is was an NO-mediated response.
Using a completely different approach, adult human fibroblasts extracted from skin biopsies were harvested from individuals with advanced cardiovascular disease. In those studies (31), tissue-engineered blood vessels served as arterial bypass grafts in long-term animal models. These tissue-engineered blood vessels had mechanical properties similar to human blood vessels, without relying upon synthetic or exogenous scaffolding. The tissue-engineered blood vessels were shown to be antithrombogenic and mechanically stable for eight months in vivo, and histological analysis showed complete tissue integration and formation of vasa vasorum. The endothelium was confluent and positive for von Willebrand factor. The process of developing this type of blood vessel differed from that originally described (29) in that the smooth muscle cell layer was excluded.
Enabling Technologies: Bioreactors
There are numerous enabling technologies that will contribute to tissue engineering and regenerative medicine (see Figure 3⇑). We will discuss bioreactors because of their particular relevance for pharmacological scientists. In fact, as one looks to the future of tissue engineering, it would seem that bioreactors, that is, laboratory devices that are capable of recapitulating relevant aspects of the in vivo environment, may be extremely useful tools for the in vitro development of isolated engineered human tissues. We will use the bioengineered vessel as an example of the potential importance of this enabling technology.
One of the final steps in creating tissue-engineered blood vessels would involve the development of a bioreactor system for cell seeding and preconditioning prior to implantation. In this regard, bioreactors provide a controllable, mechanically active environment that can be used to study and potentially improve engineered tissue structure, properties, and integration. The rationale for the use of bioreactors for tissue-engineered blood vessels derives from studies demonstrating that mechanical stress can accelerate cell and tissue growth and phenotypic differentiation (34–37). For cell seeding onto scaffolds, many studies previously employed conventional methods (e.g., static seeding in petri dishes), which typically yield engineered constructs comprising a thin tissue layer at the base of the scaffold, owing to the gravitational settling of cells. In contrast, convective mixing (e.g., in spinner flask bioreactors) and convective flow (e.g., in perfused bioreactors) can improve initial cell function. For the cultivation of engineered tissues, bioreactors can provide a more flexible environment in order to improve construct cellularity and molecular composition, as well as to permit studies of mass transport of oxygen and growth factors, hydrodynamic conditions, and physical stimuli, such as dynamic compression or cyclic stretch (38, 39). Bioreactors may also be used for pharmacological manipulation of tissues. In fact, a variety of approaches have been attempted to seed both the endothelium and smooth muscle cells, and recent published studies have documented highly evolved bioreactor systems to produce and monitor the mechanical forces required for cell seeding and preconditioning (40–47).
Implantation of more mature and functional tissue-engineered blood vessels would presumably accelerate tissue formation and maturation in vivo, thereby providing for quicker clinical results. As this technology continues to advance, one can imagine a maturation process sufficient to yield implants suitable for shipping to surgeons. In addition, it is anticipated that this technology will provide for engineered human tissues with many of the features of native tissues, thus permitting in vitro investigations for potentially prolonged periods of time.
Current Clinical Applications of Regenerative Medicine for Bladder and Vessels
Certainly, more precise details will be required concerning the physiological and pharmacological characteristics of bioengineered tissues as they traverse the pathway from in vitro manufacturing to surgical implantation in the patient, and then into the in vivo maturation process to become a functioning neo-organ. Nonetheless, the potential clinical utility of tissue engineering is beginning to be evaluated with respect to both bioengineered neobladders and blood vessels. The information thus far is scant, but the advent of these trials, described below, is exciting.
Experience with Neobladders
In patients with end-stage bladder disease, gastrointestinal tissue can be used for bladder reconstruction, but the complications associated with this process of cystoplasty can be numerous. As an alternative process, Atala and colleagues have successfully used autologous engineered bladder tissues for the reconstruction (10) of bladders in seven patients (aged four through nineteen, all with myelomeningocele) with high-pressure or poorly compliant bladders. The investigators used bladder biopsies from each patient to establish urothelial and smooth muscle cells in culture; cells were then seeded on a biodegradable bladder-shaped collagenous scaffold. About seven weeks after initial biopsy, the autologous engineered bladder constructs were used for reconstruction and implanted either with or without an omental wrap. One to five years after implantation, the engineered bladders with an omental wrap manifested superior characteristics (bladder leak point pressure decrease at capacity, volume increase, compliance increase) relative to those without omental wrap. Biopsies of the engineered bladders, furthermore, revealed adequate structural architecture and phenotype. In addition, a phase-II clinical trial of neobladders engineered according to this process has been initiated under an investigational new drug (IND) petition approved by the Food and Drug Administration (FDA) (www.Tengion.com; www.clinicaltrials.gov.) in patients with neurogenic bladders secondary to spina bifida (myelodysplasia).
Engineered Blood Vessels
Clinical trials with human blood vessels are still in their infancy. The first clinical application of an artificial vessel based on a biodegradable scaffold was reported by Shin’oka and coworkers in 2001 (48). In this report, the peripheral pulmonary artery was successfully reconstructed in a four-year-old girl with the patient’s own venous cells seeded onto a polycaprolactone–polylactic acid copolymer tube that was reinforced with woven PGA. The same investigators reported on tissue-engineered grafts seeded with autologous bone marrow cells having been implanted in forty-two patients (49). The polymer tube serving as a scaffold for the cells was composed of a copolymer of L-lactide and ε-caprolactone (50:50), which is degraded by hydrolysis. Polyglycolic acid woven fabric with a thickness of 0.5 mm was used for reinforcement. Twenty-three tissue-engineered conduits (grafts for extracardiac total cavopulmonary connection) and nineteen tissue-engineered patches were used for the repair of congenital heart defects. Upon follow-up some months to years later, there were no signs of thrombosis, stenosis, obstruction of the tissue-engineered autografts, or other complications. There was no evidence of aneurysm formation or calcification on cineangiography or computed tomography. All tube grafts were patent, and the diameter of the tube graft increased with time.
In addition, Cytograft (Novato, California) reported the results from a pilot clinical study looking at the use of engineered blood vessels in patients on hemodialysis. Grafts were implanted as arteriovenous fistulas for dialysis access and were allowed to mature in vivo before use. The vessels were constructed from autologous dermal fibroblasts and endothelial cells with the use of a sheet-based methodology over a culture period of several months. The major finding was that during up to five months of implantation, no failures were noted with the first three patients, and the grafts were functioning well for hemodialysis access. Cytograft currently has approval for two clinical trials on coronary bypass patients (www.cytograft.com).
Conclusions: the Future is Now!
To date, pharmacological sciences have been applied to regenerative medicine/tissue engineering on a goal-directed, tissue-specific basis. However, what is required to achieve the ultimate potential of these groundbreaking regenerative technologies is a more systematic application of pharmacological principles at each of the different steps in the process. Some concrete examples of how this can be accomplished are given in Table 1⇓. There is obviously enormous potential and demand for pharmacology to shape the development of regenerative medicine.
Areas of Demand for Pharmacology in Regenerative Medicine
Acknowledgments
We would like to thank Dr. James Yoo for valuable comments and suggestions.
- © American Society for Pharmacology and Experimental Theraputics 2007
References

Karl-Erik Andersson, MD, is Professor at the Wake Forest Institute for Regenerative Medicine. His broad interests include urogenital and cardiovascular pharmacology and physiology, clinical trials, and regenerative pharmacology.

George J. Christ, PhD, is Professor at the Wake Forest Institute for Regenerative Medicine. His broad interests include muscle physiology, intercellular communication, and the role of smooth muscle in the function and dysfunction of visceral and vascular tissues. Address correspondence to GJC. E-mail gchrist{at}wfubmc.edu; fax 336-713-7290.