Painful Research: Identification of a Small-Molecule Inhibitor that Selectively Targets Nav1.8 Sodium Channels
Pain is a significant medical problem, and among its many types, neuropathic pain can be difficult to treat effectively. The blockade of sodium channels, which are responsible for the rapid membrane depolarization characteristic of action potentials (APs) in neurons and for successful transmission of painful stimuli along sensory fibers, has long been known to provide pain relief. Unfortunately, the usefulness of sodium channel blockers is limited by narrow therapeutic windows resulting from, at least in part, the ability of sodium channel blockers (such as lidocaine) to inhibit the activity of cardiac and brain sodium currents in addition to the sodium currents in peripheral sensory neurons. Thus, while lidocaine can be used as a local anesthetic, it is also an antiarrhythmic and can be highly toxic when given systemically. Exciting research on sodium channels and currents in peripheral sensory neurons carried out over the last two decades has indicated that it might be possible to develop sodium channel inhibitors that selectively and effectively target pain sensation. Only within the last year, however, have sodium channel inhibitors been identified that begin to fulfill this promise (1–3).
Sodium currents in different neuronal tissues were once thought virtually identical. More recently, however, nine different genes have been identified that produce distinct voltage-gated sodium channel isoforms, seven of which are expressed in neurons (4). The different sodium channel isoforms exhibit distinct biophysical characteristics, play a variety of roles within neurons, and interact with one another to modulate firing behavior (4, 5). Although selective deployment of isoforms in different tissues and modulation by endogenous factors can provide an exquisite level of plasticity within neurons, changes in sodium channel expression and in their functional properties can lead to pathogenesis (6). Peripheral sensory neurons express complex sodium currents, with fast-inactivating components that are sensitive to the classic sodium channel blocker tetrodotoxin (TTX) and slow-inactivating sodium currents that are TTX-resistant (TTX-R). In 1996, the first neuronal TTX-R sodium channel, originally referred to as sensory neuron–specific (SNS) and now called Nav1.8, was cloned (7). In 1998, a second TTX-R neuronal sodium current, termed NaN or Nav1.9, was cloned from sensory neurons (8). Because of their predominant expression in dorsal root ganglion (DRG) neurons and associated sensory fibers (7, 8), both of these subtypes attracted significant interest as possibly providing targets for “magic bullets” to treat pain.
Several lines of evidence indicate that Nav1.8 channels, and perhaps to a lesser extent Nav1.9 channels, are important contributors to pain sensation. Work on Nav1.8 and Nav1.9 has concentrated on three main areas: 1) effects of agents on the biophysical properties of TTX-R currents; 2) alterations of channel expression and properties in models of neuropathic pain; 3) and the use of genetic knockout mice or antisense technology to explore the physiological roles of Nav1.8 and Nav1.9. Inflammatory agents were found to change the biophysical characteristics of Nav1.8 and Nav1.9 currents in ways that are likely to lead to increased excitability (9–11). Mice lacking Nav1.8 show deficits in noxious, mechanical, thermal, and visceral responses (12, 13), and delayed development of inflammatory hyperalgesia (12). Recent studies on Nav1.9 knockout mice indicate that this isoform also plays a role in inflammatory pain (14). Thus, whereas Nav1.8 and Nav1.9 appear to participate in inflammatory pain, other data from the knockout mice studies suggests that neither Nav1.8 nor Nav1.9 is essential for the development of neuropathic pain conditions (13, 14). In fact, the expression of Nav1.8 and Nav1.9 is reduced in many neuropathic pain models (15). It is important to note that results from knockout studies might be confounded by compensatory mechanisms: for instance, in one study of TTX-R knock-out mice, TTX-sensitive (TTX-S) channel expression was significantly increased (12). In contrast to the genetic knockout experiments, several studies using antisense technology or short inhibitory RNA (siRNA)-mediated knockdown approaches to decrease Nav1.8 expression appear more conclusive (16, 17); these experiments suggest that even if Nav1.8 is not responsible for the development of neuropathic pain states, it may still be a necessary component of hyperalgesia. Thus, selective and potent block of Nav1.8 may be highly desirable. Indeed, selective blockers of Nav1.8 should significantly enhance our ability to investigate the role of Nav1.8 in pain mechanisms and could potentially lead to powerful new strategies for managing pain.
A new study by Jarvis et al. reports the identification of A-803467 a small-molecule sodium channel inhibitor of Nav1.8. A-803467 is significantly more potent against recombinant human Nav1.8 channels than against several recombinant human TTX-S isoforms and the very important TTX-R cardiac isoform expressed in a heterologous cell-line (3) (Figure 1⇓). These authors investigated the antinociceptive activity of A-803467 using a variety of animal pain models, including models of acute, inflammatory, neuropathic, and visceral pain. Significant reductions in pain-related behaviors were observed in many, but not all, of the pain models. A-803467 potently reduced thermal hyperalgesia in inflammatory models of pain and mechanical allodynia in neuropathic models of pain but was relatively ineffective in a skin-incision model of postoperative pain, a chemotherapy-induced neuropathic pain model, and in models of visceral pain. An intriguing outcome from the different pain models examined is that A-803467 is inactive against acute thermal pain but is active against acute mechanical pain and cold allodynia (i.e., pain associated with a stimulus that does not cause injury). Indeed, data from Zimmerman et al. confirm that Nav1.8 is essential for transmission of painful stimuli under cold conditions (18), results that fit well with the pain-blocking profile of A-803467 (3).
Recent studies in humans with inherited complete insensitivity to pain indicates that a selective loss of the ability to sense pain can result from homozygotic mutations that eliminate functional Nav1.7 channels, indicating that Nav1.7 is a critical determinant of pain sensitivity (19). As demonstrated by Jarvis et al., A-803467 was almost 500-fold more potent at blocking recombinant Nav1.8 channels than recombinant Nav1.7 channels (3); however, these authors did not determine the sensitivity of Nav1.9 or of Nav1.6, a TTX-S channel widely expressed in DRG (20), to A-803467. An additional concern is that no recordings are shown for selectivity for Nav1.8 vs TTX-S currents in DRG neurons, which is an important consideration despite the indirect AP evidence presented. Furthermore, their data show that the block of human Nav1.8 heterologously expressed in a cell line is greater than the block observed in rat Nav1.8 and TTX-R currents in rat DRG. This finding could imply that the compound has higher affinity for human channels, versus rat channels but additional complications might involve differences arising from changes in the biophysical characteristics of sodium channels when expressed in different cell types. This question could be addressed by examining block of Nav1.8 channels expressed in DRG neurons of Nav1.8-null mice using transfection technology. Additionally, whereas the DRG recordings by Jarvis et al. were completed with TTX present, experiments by these authors utilizing the human Nav1.8 cDNA in cell lines were conducted in the absence of TTX. Very high concentrations of TTX can block TTX-R currents, and if the A-803467 binding site is close to that of TTX site, then A-803467’s efficacy could be affected.
In contrast to many sodium channel blockers in clinical use, A-803467 does not appear to block channels in a use-dependent manner. Use-dependent block refers to the additional block that occurs when channels are activated (depolarized) at relatively high frequencies. Jarvis et al. compared A-803467 to the use-dependent blockers tetracaine and mexiletine and found that unlike those standard compounds, high frequency trains of depolarization did not increase the block caused by A-803467 (3). This is important, as use-dependent block has been proposed to be a critical part of the therapeutic action of some sodium channel blockers. Inhibiting abnormal higher-frequency activity—for example, high-frequency activity associated with neuropathic pain—while allowing normal low-frequency activity to continue might increase therapeutic effectiveness and simultaneously limit side effects. The lack of use-dependence could explain why A-803467 is less effective in some pain models than others; however, the data did show that although A-803467 does block TTX-R channels at rest, the block is significantly greater at more depolarized potentials. This finding indicates that A-803467 possesses a greater affinity for the inactivated state of the channel, an observation noted with other clinically useful sodium channel inhibitors. Interestingly, when A-803467 was removed, the block was reversed (except at depolarized voltages), reinforcing the idea that the drug preferentially binds to the inactivated states. In inflammatory and neuropathic pain conditions, prolonged depolarizations are likely to occur; therefore, the preferential interaction of A-803467 with inactivated states of Nav1.8 is likely to play an important role in the mechanism of analgesic action. Indeed, Jarvis et al. studied APs from isolated neurons and although they report a lack of block of APs under normal conditions by A-803467, they show a robust A-803467-mediated block of AP trains from depolarized potentials, whether artificially induced or stemming from treatment with complete Freund’s adjuvant (3). The APs from these depolarized potentials are likely to be conducted predominantly by Nav1.8 channels, but it would have been useful to test the effects of A-803467 on TTX-R spikes alone and to analyze the AP more thoroughly. For instance, as a result of slower kinetics (4, 7), APs involving Nav1.8 tend to be broader than those associated with TTX-S channels; thus, the duration of Nav1.8-dependent APs should be reduced in the presence of the blocker.
Although there are differences in the effectiveness of A-803467 in the various pain models, the data in Jarvis et al. point towards Nav1.8 being an extremely viable target for effective pain relief in inflammatory and, importantly, neuropathic pain. Recent studies on humans with complete insensitivity to pain indicate that Nav1.7-selective blockers might act as ideal analgesics (19); however, there is compelling evidence that the coordinated activity of Nav1.7 and Nav1.8 may be necessary for nociceptor excitability (5); therefore, blocking either of these subtypes should result in pain relief. A peptide conotoxin identified in marine snails, MrVIb, also has inhibitory activity against Nav1.8 (1, 2), but MrVIb exhibits only approximately tenfold higher selectivity for Nav1.8 over other neuronal sodium currents and only about two-fold higher selectivity for Nav1.8 over the skeletal muscle sodium channel Nav1.4, which could limit the usefulness of MrVIb (2). In contrast, A-803467 exhibits at least 100-fold higher affinity for Nav1.8 over the other subtypes that it was tested against, and, as a small molecule inhibitor, A-803467 may have greater therapeutic potential as compared to peptide inhibitors. Additional experiments such as those suggested above are needed to further explore the effectiveness of A-803467 as a selective blocker of Nav1.8. The chemistry surrounding this compound should provide new avenues for finding additional selective blockers of Nav1.8, which would give further evidence for the validity of these compounds as future drug candidates for pain treatment and also provide extremely valuable experimental tools for investigators researching the role of TTX-R Nav1.8 channels in nociception.
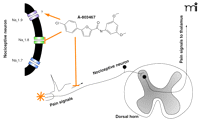
Inhibiting Nav1.8-dependent nociception. The small molecule A-803467 potently inhibits Nav1.8 tetrodotoxin-resistant (TTX-R) channels in nociceptive neurons. A-803467 also inhibits action potentials in depolarized neurons or spontaneously active neurons following peripheral inflammation. A-803467 is relatively ineffective at inhibiting Nav1.7 channels, which are now known to participate in nociception. It is not known, however, whether A-803467 can inhibit Nav1.9, another TTX-R sodium channel that is expressed in large amounts in nociceptive neurons and can contribute to inflammatory pain. Orange star indicates pain stimulus.
- © American Society for Pharmacology and Experimental Theraputics 2007
References

Theodore R. Cummins is an Assistant Professor of Pharmacology & Toxicology at Indiana University School of Medicine. He received his PhD in Neuroscience from Yale University, working with Fred Sigworth. He then trained with Stephen Waxman in the Department of Neurology at Yale University. Dr. Cummins’s laboratory in Indianapolis focuses on sodium channel pharmacology and the role of sodium channels in disorders of excitability. Address correspondence to TRC. E-mail: trcummin{at}iupui.edu; fax 317-278-5849.

Tony Rush received his PhD from Dundee University under the guidance of Jim Elliott, characterizing sodium channels of DRG neurons. He then moved to Trinity College Dublin to study plasticity with Roger Anwyl before continuing sodium channel research with Stephen Waxman at Yale University. He is currently a Senior Research Scientist with NeuroSolutions Ltd, UK providing electrophysiological services to the biotechnology and pharmaceutical industries. E-mail: TRush{at}neurosolutionsltd.com