|
 
 |
|
ORIGINAL ARTICLE |
|
|
|
| Year : 2014 | Volume
: 20
| Issue : 1 | Page : 59-63 |
| |
Association of single nucleotide polymorphisms of CACNA1A gene in migraine
Aadil Bashir1, Shiekh Saleem1, Maqbool Wani1, Roohi Rasool2, Irfan Yousuf Wani1, Azhara Gulnar1, Sawan Verma1
1 Department of Neurology, SKIMS, Soura, Srinagar, Jammu and Kashmir, India
2 Department of Immunology, SKIMS, Soura, Srinagar, Jammu and Kashmir, India
| Date of Web Publication | 19-May-2014 |
Correspondence Address:
Irfan Yousuf Wani
Department of Neurology, SKIMS, Soura, Srinagar, Jammu and Kashmir
India
 Source of Support: Indian Council of Medical Research, Conflict of Interest: None
DOI: 10.4103/0971-6866.132757

 Abstract Abstract | | |
Introduction: Migraine is a chronic, neurovascular polygenic disease where genetic and environmental factors are involved in its etiology. Dysfunction of neuronal ion transportation can provide a model for predisposition for common forms of migraine. Mutations in genes encoding ion channels disturb the rhythmic function of exposed tissue that may also explain the episodic nature of migraine. Our aim was to study the single nucleotide polymorphisms of CACNA1A gene in migraine patients.
Materials and Methods: The subjects were the patients of migraine, in the age range of 18-80 years, diagnosed by a Neurologist, as per the diagnostic criteria of International Headache Society (IHS) Classification 2004 after excluding other causes of headache by clinical examination and relevant investigations.
The controls were the age and sex matched healthy persons from the same population excluding the relatives of patients. Only those patients and the controls, who voluntarily participated in the study, were taken and their blood samples were taken for the study. Deoxyribonucleic acid (DNA) extraction was performed according to the manufacturer's protocol for Qiagen DNA extraction kits (Qiagen, Hilden, NRW, Germany). DNA content was quantified by spectrophotometric absorption (Nanodrop Spectrophotometer, BioLab, Scoresby, VIC, Australia). Polymerase chain reaction was performed using an iCycler Thermal Cycler (Bio-Rad, Hercules, CA, USA). The polymorphic analysis of CACNA1A gene was carried out by two methods: Restriction fragment length polymorphism and sequencing.
Results: The study included a total of 25 patients of migraine, diagnosed on out-patient department basis as per IHS Classification 2004 and compared with age and sex matched 25 healthy controls. Most of the patients 23 (92%) were below the age of 50 years. 20 of the patients (80%) were females and 5 (20%) were males. The polymorphic analysis of CACNA1A gene revealed the presence of only the wild form of the gene for the codon E993V in both case and control groups.
Conclusion: In our study, we could not find any polymorphism of CACNA1A gene in the selected patients. Instead the wild type of genotype was found in both patients and controls. This negative result presented here, implies that if the CACNA1A gene is involved in typical migraine (with and without aura), its contribution is very modest and therefore difficult to discern. Nevertheless, there are other genes that could be considered potential candidates for typical migraine susceptibility for which further research is needed.
Keywords: CACNA1A gene, migraine, polymorphism
How to cite this article:
Bashir A, Saleem S, Wani M, Rasool R, Wani IY, Gulnar A, Verma S. Association of single nucleotide polymorphisms of CACNA1A gene in migraine. Indian J Hum Genet 2014;20:59-63 |
How to cite this URL:
Bashir A, Saleem S, Wani M, Rasool R, Wani IY, Gulnar A, Verma S. Association of single nucleotide polymorphisms of CACNA1A gene in migraine. Indian J Hum Genet [serial online] 2014 [cited 2016 Aug 24];20:59-63. Available from: http://www.ijhg.com/text.asp?2014/20/1/59/132757 |
 Introduction Introduction | |  |
Migraine is a chronic, neurovascular polygenic disease where genetic and environmental factors are involved in its etiology. It affects about 15% of the population. There is a higher prevalence in women (18%) when compared with men (6%), with attacks lasting from a few hours to days. [1] Onset of migraine is nearly always below age 50 (in 90% of patients). Peak age of onset of migraine is 10-12 years old for males and 14-16 years old for females. [2],[3]
Diagnosis of migraine is based on the patient's description of the attacks and exclusion of other possible secondary causes of headache. Based on the International Headache Society (IHS) Classification, [4] migraine with aura (MA) and without aura (MO), are the two most frequent forms of this disease (20-30% MA, 70-80% MO). [5]
Etiology of a migraine attack is only partly understood. Environmental influences play a role, but family studies suggest that defective genes may be primarily involved in disease causation. The mode of transmission of typical migraine in families is still unclear, but it is widely believed to be multifactorial, [6] although a role for a major susceptibility gene cannot be excluded. [7]
So far, genetic studies have not identified susceptibility variants for common forms of migraine, i.e. MA and MO. Due to similarities and co-occurrence between common migraine and familial hemiplegic migraine (FHM), dysfunction of neuronal ion transportation can provide a model for predisposition for common forms of migraine. Mutations in genes encoding ion channels disturb the rhythmic function of exposed tissue that may also explain the episodic nature of migraine. [8],[9]
 Aims and Objectives Aims and Objectives | |  |
The aim of the following study is to determine the single nucleotide polymorphisms (SNPs) of CACNA1A gene in migraine patients following Neurology Out-patient Department at SKIMS Srinagar.
 Materials and Methods Materials and Methods | |  |
Materials
The subjects were the patients of migraine, in the age range of 18-80 years, diagnosed by a neurologist, as per the diagnostic criteria of IHS Classification 2004 [4] after excluding other causes of headache by clinical examination and relevant investigations such as brain imaging, angiography, electroencephalography and cerebrospinal fluid analysis.
The controls were the age and sex matched healthy persons from the same population excluding the relatives of patients. Only those patients and the controls, who voluntarily participated in the study, were taken and their blood samples were taken for the study after permission from the Institute Ethical Clearance Committee.
Methods
All the participants were informed and consent was taken for participation in the study. A volume of 5 ml peripheral blood samples were collected in ethylenediaminetetraacetic acid vials from patients and healthy controls and later stored at −80°C until further use. Out of the 25 cases, 5 were males and 20 females with the age distribution of ≤50 years, 23 and >50 years. [2] The control group had 9 males and 16 females with the age distribution ≤50 years, 21 and >50 years. [4]
CACNA1A genotyping
Deoxyribonucleic acid (DNA) extraction was performed according to the manufacturer's protocol for Qiagen DNA extraction kits (Qiagen, Hilden, NRW, Germany). DNA content was quantified by spectrophotometric absorption (Nanodrop Spectrophotometer, BioLab, Scoresby, VIC, Australia). Polymerase chain reaction (PCR) was performed using an iCycler Thermal Cycler (Bio-Rad, Hercules, CA, USA). For CACNA1A E993V A > T (rs16023) genotype was determined using PCR-restriction fragment length polymorphism (RFLP) method. Amplifications were carried out and a PCR product was amplified using the specific forward and reverse primer. Primers were designed and selected using Primer3, version 0.4.0 software (Whitehead Institute for Biomedical Research). For CACNA1A E993V A > T (rs16023), the primers were forward 5'-ACC TCG AGT GCA GTC GTC CAG CCC GAG GCG GAG AG-3'reverse 5'- CCT CCT CCG ATG CCT CCG CTC CTT GTC CTC CCT CC-3'. The PCR reaction mixture consisted of Taq (1.5 U), sense and antisense primers (0.5 μmol/l), Mg 2+ (50 mmol/l), deoxyribonucleotide triphosphate (0.2 mmol/l) and template (1 μg) and was subjected to an initial denaturing step of 4 min at 95°C, then 35 cycles of denaturing for 30 s at 95°C, annealing for 30 s at 65°C, extension for 30 s at 72°C and a final extension step of 10 min at 72°C. Digestion of the amplified products of E993V A > T was done by using 10 units restriction endonucleases BsaX1I (fermentas) and incubated at 37°C for 16 h. The digested products were checked on 3% agaroses gel, the RFLP picture for E993V genotype was identified as 100, 52, 33 and 19 bp in the presence of the V993 allele i.e. four bands were seen which could have occurred due to the presence of more than one restriction site. RFLP results were later confirmed by sequencing at Macrogen Korea.
 Results Results | |  |
This study was conducted to see the association between migraine and CACNA1A gene polymorphism. The study included a total of 25 patients of migraine, diagnosed on out-patient department (OPD) basis as per IHS Classification 2004 and compared with age and sex matched 25 healthy controls. Most of the patients 23 (92%) were below the age of 50 years. Twenty of the patients (80%) were females and 5 (20%) were males [Table 1].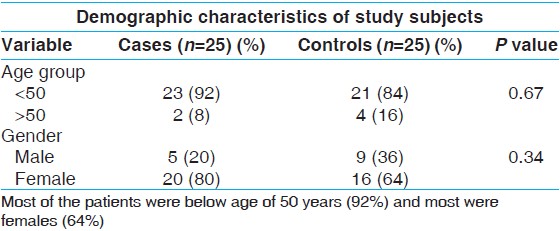 | Table 1: Representing the demographic characteristics of patients and controls
Click here to view |
Out of 25 patients, 21 (84%) were having MO and 16 (64%) had a positive family history [Table 2]. With regard to frequency of attacks, 3 (12%) were having weekly, 9 (36%) had 2-3 weekly, 8 (32%) had 4-6 weekly and 5 (20%) were having attacks >6 weekly. The associated features including nausea/vomiting, photophobia and phonophobia were present in 20 (80%), 16 (64%) and 12 (48%) patients respectively. With regard to severity, 6 (24%) had mild, 12 (48%) had moderate and 7 (28%) had severe migraine. Out of the total patients 18 people were experiencing headache lasting < 2 days and 7 were having attacks lasting > 2 days.
The polymorphic analysis of CACNA1A gene carried out by two methods: RFLP and sequencing, revealed the presence of only the wild form of the gene for the codon E993V in both case and control groups.
The above table shows that maximum patients (84%) were without aura nausea/vomiting was the most common associated symptom (80%) followed by photophobia (64%) and phonophobia (48%).
Genomic DNA extraction was done by standardized protocols in our laboratory in immunology and molecular medicine. Quality of genomic was checked on 1% agarose gel. The gel was visualized under ultraviolet (UV) transilluminator.
Amplification of the target region was carried out by using forward and reverses primer in thermal cycler (Bio Rad). PCR program was 95°C for 7 min, followed by 35 cycles of 95°C for 35 s, 65°C for 35 s, 72°C and final extension of 72°C for 7 min. The amplified products was loaded on 2% agarose gel along with 100 bp marker and was later checked under UV transilluminator as given in [Figure 1].
Sequencing of the amplified product was carried out at Macrogen (Korea).
The results obtained revealed the presence of only wild type of genotype in both cases and controls [Table 3].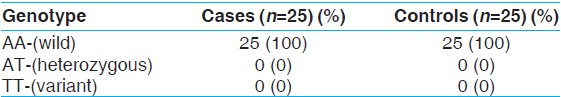 | Table 3: Genotype frequencies CACNA1A gene polymorphism in cases and controls
Click here to view |
 Discussion Discussion | |  |
Due to the complexity of typical migraine, the identification of causative genes has been difficult. However, in 1996, FHM was shown to be caused by four different missense mutations in conserved functional domains in the calcium channel gene CACNA1A located on 19p13. [10] The CACNA1A gene, which encodes the main subunit of the P/Q type neuronal calcium channel [10],[11],[12],[13] has been implicated in a percentage of patients presenting with FHM1. [14],[15],[16] CACNA1A encodes the pore-forming alpha 1 subunit of neuronal CaV2.1 (voltage-gated P/Q-type Ca++) channels. This subunit consists of four repeated domains (I-IV), each of which contains six transmembrane regions (S1-S6) comprising a voltage sensor S4 and a pore loop between S5 and S6 respectively. [17] FHM is a rare and genetically heterogeneous autosomal dominant subtype of MA. Mutations in the genes CACNA1A have been identified and correlated to the FHM. [5] FHM1 mutations produce gain-of-function of the Ca (V) 2.1 channel and as a consequence, increased Ca (V) 2.1-dependent neurotransmitter release from cortical neurons and facilitation of in vivo induction and propagation of cortical spreading depression the phenomenon underlying migraine aura. [18] According to International Classification of Headache Disorders-II, basilar-type migraine (BM) only differs from HM by the lack of hemiplegia. This suggests that HM and BM may have a common genetic substrate, which is supported by a recent finding of a mutation in ATP1A2 in a family with BM. [19]
The search for genes involved in the pathophysiology of migraine poses major difficulties. First, there is no objective diagnostic method to assess the status of the individuals studied. Second, migraine is a polygenic multifactorial disorder. In addition to genetic factors, familial aggregation of a pathological disorder may be due to environmental factors and may also, as previously mentioned, occur purely by chance in very common diseases. Genetic epidemiological surveys for migraine genetics showed the existence of genetic factors. A Danish twin study was carried out in a large population using the IHS diagnostic criteria, included 1013 monozygotic and 1667 dizygotic twin-pairs, the pairwise concordance rate was significantly higher among monozygotic than dizygotic twin-pairs for MO (28 vs. 18%, P < 0·05) and for MA (34 vs. 12%, P < 0·001). [5]
D'Onofrio et al. in 2009 [20] did a study regarding the interplay of two SNPs in the CACNA1A gene and migraine susceptibility wherein the E918D and E993V SNPs were found in 30/117 (25.6%) and 32/117 (27.3%) migraine patients, respectively. The prevalence of these SNPs taken separately was not significantly different from that of control subjects (n = 28/107, 26.2% for E918D; n = 29/107 for E993V, 27.1%) neither for the total migraine population nor for the various migraine subtypes. By contrast, co-existence of both SNPs was more frequent in migraineurs (25/117, 21%) than in healthy controls (12/107, 11%; P = 0.048), a difference that was significant for every migraine subtype. This result suggests that the interplay of minor genetic variants such as SNPs may influence the P/Q-type calcium channel function in several subtypes of migraine. [20]
Gerola et al. did a case-control association study in a cohort of Italian population, over 18 years of age, to assess whether two common non-synonymous SNPs in the CACNA1A gene may represent predisposing factors for MA or MO. The study suggested that rs16022 and rs16027 CACNA1A variants did not represent primary predisposing factors for the pathogenesis of common migraine in Italian patients. However, since two SNPs cannot describe the entire gene variability, it could not exclude a role of CACNA1A in common migraine. [21]
CACNA1A gene mutations have been implicated in spinocerebellar ataxia (specifically SCA6) [21] and in episodic ataxia type 2. [10],[22],[23],[24],[25]
The minor allele frequencies of this variant in world populations is A = 0.124/271. [26]
Our study was carried in a Kashmiri population which is an ethnically related to Aryans. In this population there is greater incidence of consanguineous marriage. In our study after analyzing the samples of patients and controls for the polymorphism of CACNA1A gene, we could not find any such polymorphism in the selected patients, of Kashmiri population. Instead the wild type of genotype was found in both patients and controls. This negative result presented here, implies that if the CACNA1A gene is involved in typical MA and MO, its contribution is very modest and therefore difficult to discern. Nevertheless, there are other genes that could be considered potential candidates for typical migraine susceptibility, which needs continuous research.
 References References | |  |
| 1. | Kors E, Haan J, Ferrari M. Migraine genetics. Curr Pain Headache Rep 2003;7:212-7. 
|
| 2. | Stewart WF, Shechter A, Rasmussen BK. Migraine prevalence. A review of population-based studies. Neurology 1994;44 6 Suppl 4:S17-23. 
|
| 3. | Abu-Arefeh I, Russell G. Prevalence of headache and migraine in schoolchildren. BMJ 1994;309:765-9. 
|
| 4. | Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2 nd edition. Cephalalgia 2004;24 Suppl 1:9-160. 
[PUBMED] |
| 5. | Ducros A, Tournier-Lasserve E, Bousser MG. The genetics of migraine. Lancet Neurol 2002;1:285-93. 
|
| 6. | Russell MB, Olesen J. The genetics of migraine without aura and migraine with aura. Cephalalgia 1993;13:245-8. 
|
| 7. | Mochi M, Sangiorgi S, Cortelli P, Carelli V, Scapoli C, Crisci M, et al. Testing models for genetic determination in migraine. Cephalalgia 1993;13:389-94. 
|
| 8. | Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry 2006;60:177-85. 
[PUBMED] |
| 9. | Bernard G, Shevell MI. Channelopathies: A review. Pediatr Neurol 2008;38:73-85. 
|
| 10. | Ophoff RA, Terwindt GM, Vergouwe MN, van Eijk R, Oefner PJ, Hoffman SM, et al. Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+channel gene CACNL1A4. Cell 1996;87:543-52. 
|
| 11. | Fumal A, Schoenen J. Genetics of migraines: From ionic channels to single nucleotide polymorphisms? Rev Med Liege 2004;59:367-77. 
|
| 12. | Jen JC, Kim GW, Dudding KA, Baloh RW. No mutations in CACNA1A and ATP1A2 in probands with common types of migraine. Arch Neurol 2004;61:926-8. 
|
| 13. | Kim EY, Rumpf CH, Fujiwara Y, Cooley ES, Van Petegem F, Minor DL Jr. Structures of CaV2 Ca2+/CaM-IQ domain complexes reveal binding modes that underlie calcium-dependent inactivation and facilitation. Structure 2008;16:1455-67. 
|
| 14. | Pietrobon D. Familial hemiplegic migraine. Neurotherapeutics 2007;4:274-84. 
[PUBMED] |
| 15. | Cao YQ, Tsien RW. Effects of familial hemiplegic migraine type 1 mutations on neuronal P/Q-type Ca2+channel activity and inhibitory synaptic transmission. Proc Natl Acad Sci U S A 2005;102:2590-5. 
|
| 16. | Moskowitz MA, Bolay H, Dalkara T. Deciphering migraine mechanisms: Clues from familial hemiplegic migraine genotypes. Ann Neurol 2004;55:276-80. 
|
| 17. | Catterall WA. Structure and regulation of voltage-gated Ca2+channels. Annu Rev Cell Dev Biol 2000;16:521-55. 
[PUBMED] |
| 18. | Hadjikhani N, Sanchez Del Rio M, Wu O, Schwartz D, Bakker D, Fischl B, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci U S A 2001;98:4687-92. 
|
| 19. | Ambrosini A, D'Onofrio M, Grieco GS, Di Mambro A, Montagna G, Fortini D, et al. Familial basilar migraine associated with a new mutation in the ATP1A2 gene. Neurology 2005;65:1826-8. 
|
| 20. | D'Onofrio M, Ambrosini A, Di Mambro A, Arisi I, Santorelli FM, Grieco GS, et al. The interplay of two single nucleotide polymorphisms in the CACNA1A gene may contribute to migraine susceptibility. Neurosci Lett 2009;453:12-5. 
|
| 21. | Gerola S, Battistini S, Stenirri S, Nicolodi M, Arnetoli G, Canova S, et al. CACNA1A gene non-synonymous single nucleotide polymorphisms and common migraine in Italy: A case-control association study with a micro-array technology. Clin Chem Lab Med 2009;47:783-5. 
[PUBMED] |
| 22. | Zhuchenko O, Bailey J, Bonnen P, Ashizawa T, Stockton DW, Amos C, et al. Autosomal dominant cerebellar ataxia (SCA6) associated with small polyglutamine expansions in the alpha 1A-voltage-dependent calcium channel. Nat Genet 1997;15:62-9. 
|
| 23. | Spacey SD, Hildebrand ME, Materek LA, Bird TD, Snutch TP. Functional implications of a novel EA2 mutation in the P/Q-type calcium channel. Ann Neurol 2004;56:213-20. 
|
| 24. | Wan J, Carr JR, Baloh RW, Jen JC. Nonconsensus intronic mutations cause episodic ataxia. Ann Neurol 2005;57:131-5. 
|
| 25. | Riant F, Mourtada R, Saugier-Veber P, Tournier-Lasserve E. Large CACNA1A deletion in a family with episodic ataxia type 2. Arch Neurol 2008;65:817-20. 
|
| 26. | Available from: http://www.ncbi.nlm.nih.gov/snp/?term=rs16023 [Last accessed on 2014 Mar 25]. 
|
[Figure 1]
[Table 1], [Table 2], [Table 3]
|