|


 |
| CASE REPORT |
|
| Year : 2012 | Volume
: 4
| Issue : 1 | Page : 46-49 |
|
|
Functional paraganglioma: a rare conus-cauda lesion
Vivek Agrawal1, Mally Rahul1, Shadma Khan1, Velho Vernon1, Binayke Rachana2
1 Department of Neurosurgery, Grant Medical College and Sir J. J. Group of Hospitals, Mumbai, Maharashtra, India
2 Department of Pathology, Grant Medical College and Sir J. J. Group of Hospitals, Mumbai, Maharashtra, India
| Date of Web Publication | 5-Sep-2012 |
Correspondence Address:
Vivek Agrawal
Assistant Professor, Department of neurosurgery, Grant Medical College and Sir J. J. Group of Hospitals, Mumbai - 400 008
India
 Source of Support: None, Conflict of Interest: None
DOI: 10.4103/2006-8808.100355

 Abstract Abstract | | |
Conus-cauda syndrome is caused due to involvement of the lower end of the spinal cord and arising bunch of nerve roots. It is caused commonly due to traumatic injury, spinal stenosis, spinal tumors, inflammatory, and infectious conditions, but paraganglioma is a rare cause. These tumors are rarely functional and secrete catecholamine. Till now only five case reports of functional spinal paragangliomas are available to the best of our knowledge. We report a 50-year-old hypertensive male patient with a lobulated lesion extending from lower border of D12 to L2, which was reported as ependymoma on imaging studies done preoperatively. This lesion was confirmed to be a functional paraganglioma postoperatively after the patient died because of its furious complication, thus highlighting the importance of its preoperative diagnosis and management. In conclusion conus-cauda functional paragangliomas are very rare entity. Diagnosing them in preoperative condition is critical from the therapeutic point of view, both medical and surgical. During surgery these tumors should be handled very gently to avoid spillage of catecholamines into blood. These tumors require assistance of expert anesthetist and endocrinologist in the perioperative period. Keywords: Conus-cauda, catecholamine, functional, paraganglioma
How to cite this article:
Agrawal V, Rahul M, Khan S, Vernon V, Rachana B. Functional paraganglioma: a rare conus-cauda lesion. J Surg Tech Case Report 2012;4:46-9 |
How to cite this URL:
Agrawal V, Rahul M, Khan S, Vernon V, Rachana B. Functional paraganglioma: a rare conus-cauda lesion. J Surg Tech Case Report [serial online] 2012 [cited 2016 Jun 10];4:46-9. Available from: http://www.jstcr.org/text.asp?2012/4/1/46/100355 |
 Introduction Introduction | |  |
Conus-cauda syndrome is caused due to involvement of the lower end of the spinal cord and bunch of nerve roots arising from it. It can lead to a number of symptoms like low back pain, saddle sensory changes, sciatica, bladder, bowel incontinence and lower extremity motor and sensory loss. It is caused commonly due to traumatic injury, disk herniation, spinal stenosis, spinal tumors, infectious conditions, and accidental causes by medical intervention (iatrogenic causes).
Extra-adrenal pheochromocytoma is called paragang-lioma. [1],[2] It is a neuro-endocrine tumor which has a neuroectodermal origin and derives from the embryonic sympathetic and parasympathetic nervous system. [3],[4],[5],[6] The first authors to describe this pathological entity in spine were Miller and Torack in 1970, denominating it a secretory ependymoma, whereas Lerman was the first to coin the term paraganglioma of the cauda equina in 1972. [3],[4] Paragangliomas of the spine comprise 3-4% of all spinal tumors but the cases of functional paraganglioma of spine are extremely rare and only five case reports are available in the literature to the best of our knowledge. [2],[7],[8],[9] Tumors affecting the carotid body and glomus region are usually parasympathetic whereas the spinal variety are typically sympathetic. [1] We are presenting a rare case of functional paraganglioma of conus-cauda and its clinical consequences.
 Case Report Case Report | |  |
Our patient, a 50-year-old man, a known case of hypertension, on two antihypertensive drugs, was admitted with history of progressively increasing weakness with stiffness and numbness in both lower limbs since 9 months. The patient also had history of urinary and fecal incontinence. Neurological evaluation revealed spastic paraparesis with power 3/5 (MRC grading system) and decreased sensation below L1 dermatome bilaterally. Perianal sensation and anal tone were decreased. Magnetic resonance imaging of the lumbosacral spinal segment, before and after gadolinium administration, demonstrated a lobulated lesion extending from lower border of D12 to L2 vertebra level, intimately related to conus medullaris. It was isointense on T1W images [Figure 1], heterogeneous on T2W images [Figure 2]a and b and showed intense postcontrast enhancement [Figure 3]. Significant scalloping of posterior margin of vertebral bodies and their posterior elements were seen [Figure 4]. There was extension of the lesion into bilateral L1-2 neural foramina. The patient was planned for surgery keeping the possibility of ependymoma. D12 to L2 laminectomy with subtotal excision of the tumor was done. The tumor was hyper-vascular. The rostral part of the tumor was adherent to conus and was inseparable, so left behind. During surgery, blood pressure increased considerably on few occasions, but was controlled. On postoperative day 1 the patient had flushing all over the body, specially over the face and chest region, palpitation, dysphagia, and uncontrolled blood pressure. The patient was shifted to the intensive care unit where his blood pressure was not controlled in spite of giving three antihypertensive drugs. His blood and urine were sent for all necessary investigation including the serum catecholamine level. On the same day the patient collapsed and expired after an episode of hematemesis. The histopathological examination revealed a diagnosis of paraganglioma. Typical "Zellballen nests" of neoplastic cells were present surrounded by a delicate fibro-vascular stroma. [Figure 5]a and b The cells possessed granular eosinophilic cytoplasm and demonstrated nuclear pleomorphism. Immunohistochemical testing showed a positive reaction for chromogranin and S100 protein. Serum noradrenaline level was 2951 pg/ml (normal 100-450 pg/ml) with normal adrenaline level.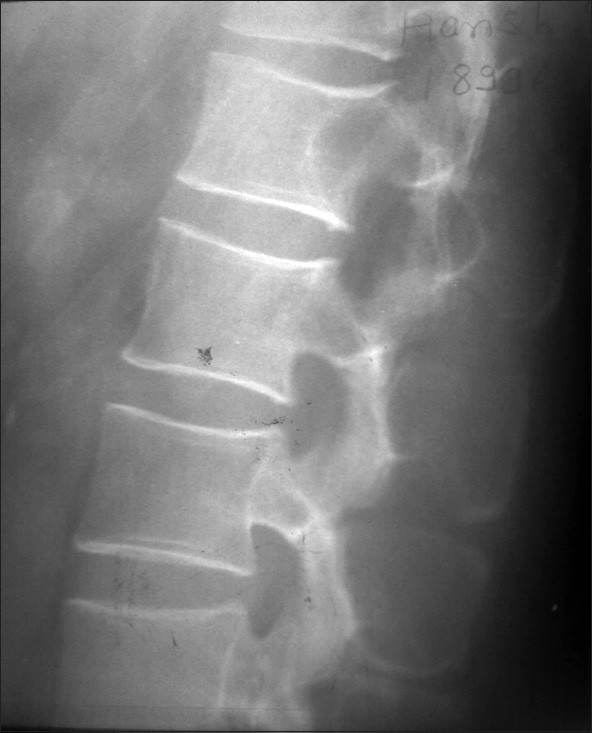 | Figure 4: X‑ray of the Dorso‑lumbar region showing significant
scalloping of posterior margin of L1 vertebral bodies and their posterior
elements
Click here to view |
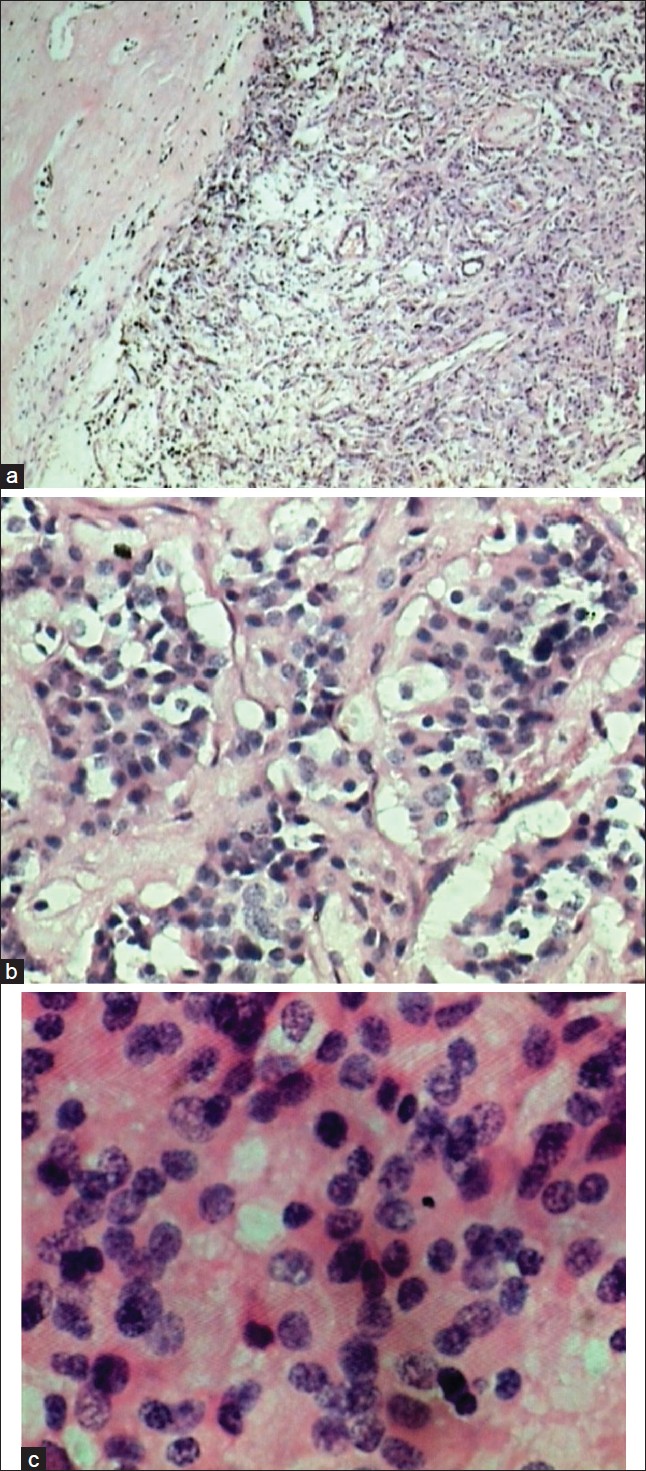 | Figure 5: (a) Squash slide; (b) Histopathological slides: suggestive "Zellballen nests" of neoplastic cells surrounded by a delicate fibrovascular stroma; Spinal paraganglioma ((H&E, 400×) showing uniform round cells with 'salt and pepper' chromatin)
Click here to view |
 Discussion Discussion | |  |
Functional paragangliomas are very rare and make about 1-3% of all extra-adrenal paragangliomas. Similarly spinal paragangliomas are mostly nonsecretory and only five case reports of secretory variety are available in the literature to the best of our knowledge [2],[7],[8],[9] [Table 1].
Functional adrenal and extradrenal paragangliomas are important but rare causes of hypertension, accounting for approximately 0.5% of the hypertensive population. [6] Management of hypertension without properly diagnosing the underlying functional paraganglioma in this group of hypertensive patients may be fatal, like in our case. In contrast, prompt diagnosis and appropriate surgical treatment are associated with 5-year survival rates of approximately 95%. [6] The diagnosis of functional paraganglioma is made on the basis of measurements of the levels of catecholamines and their metabolites in plasma or urine. The sensitivity of plasma metanephrine levels for the detection of pheochromocytoma has been reported to be as high as 100%, compared with 82% for urinary and plasma catecholamine levels. [10]
Preoperative diagnosis of functional paraganglioma is crucial from the therapeutic point of view as at least 2 weeks of α-adrenergic receptor blockade with phenoxybenzamine is required to allow the chronically contracted extravascular space to expand. [6],[10],[11] Management of hypertension with β-adrenergic receptor blockade alone is contraindicated, because such treatment can cause a paradoxical increase in blood pressure attributable to the inhibition of peripheral β2 receptors as it happened in our case. [1] Our patient was on β-blocker and angiotensin receptor blocker (ARB) in the preoperative period and was started on nitroglycerine drip in postoperative state.
The role of MRI in the diagnosis and treatment of this condition is paramount, owing to the abundance of anatomical information it provides, although a correct diagnosis may pose some problems. On T1-weighted images, the lesion usually has an isointense appearance, whereas it is hyper-intense on T2-weighted sequences with enhancement after Gadolinium administration. These features are common to other intradural lesions, such as schwannomas, ependymomas, meningiomas, metastatic tumors. In the diagnostic work-up of paraganglioma, two observations may be helpful in differentiation form other lesions (1) a defect in the serpiginous structure between the conus and the lesion, suggesting dilatation of the serpentine vessels, which is uncommon in schwannomas and ependymomas [Figure 2]a and [Figure 3]. (2) A hypointense appearance of the tumor rim on T2-weighted sequences, suggesting paramagnetic effects caused by hemosiderin, which is typical of vascular tumors, since hemosiderin discoloration is a sign of prior hemorrhage. [12],[13],[14]
Currently, the functional imaging method preferred for localizing extra-adrenal or adrenal paragangliomas is scintigraphy with metaiodobenzylguanidine (I-MIBG). Although there can be no doubt about the high level of sensitivity and specificity of this method, it also has some drawbacks. These consist of relatively high levels of radiation exposure and failure to identify nonsecreting paragangliomas.
Surgical treatment aims at complete excision of the lesion which is usually well encapsulated. However, in our case, there was breech in capsule at multiple places and the tumor was adherent to conus, so complete excision was not possible. Gentle surgical techniques and expert anesthesia are required to minimize intraoperative hypertension attributable to handling of the tumor in secreting cases. Laminectomy and dura opening while exposure of functional paraganglioma should be without compression on the tumor. Early internal debulking without dissecting the capsule and devascularization should be avoided as it may result in spillage of catecholamine so dissection outside the tumor capsule and devascularization is advisable. Excision of these tumors necessitates close cooperation between the surgeon, anesthetist, and endocrinologist in the perioperative period.
Histopathological diagnosis is based on characteristics typical of paraganglioma, namely an organoid or "Zellballen" organization of argyrophilic cells which are circumscribed by vascular stroma, immunohistochemical testing with a positive reaction for chromogranin and sustentacular cell immune reaction for S100 protein. [14],[15]
Prognosis is better in lesions that are well encapsulated. When complete removal is not feasible, radiotherapy may be considered, although resistance to this form of treatment has been documented. [16],[17] As far as chemotherapy is concerned, at the time being it does not appear to play a role in the management of these lesions. The median free interval between surgical removal and local recurrence is estimated to be approximately 6 years. [18]
 Summary Summary | |  |
We have presented a case of functional paraganglioma of the conus-cauda region. It is a very rare entity. Diagnosing it in the preoperative condition is critical as patients have to be started on a-blocker medication at least 2 weeks prior to surgery. During surgery, these tumors should be handled gently, extracapsular, with devascularization to avoid spillage of catecholamines into blood. These tumors require assistance of expert anesthetist and endocrinologist in the perioperative period.
 Conclusion Conclusion | |  |
Functional paragangliomas are extremely rare tumors of the conus-cauda region but diagnosing them preoperatively is crucial from the therapeutic point of view.
 References References | |  |
| 1. | Landi A, Tarantino R, Marotta N, Rocco P, Antonelli M, Salvati M, et al. Paraganglioma of the filum terminale: Case report. World J Surg Oncol 2009;11:95. 
|
| 2. | Jeffs GJ, Lee GY, Wong GT. Functioning paraganglioma of the thoracic spine: Case report. Neurosurgery 2003;53:992-4. 
[PUBMED] |
| 3. | Miller CA, Torack RM. Secretory ependymoma of the filum terminale. Acta Neuropathol 1970;15:240-50. 
[PUBMED] |
| 4. | Lerman RI, Kaplan ES, Daman L. Ganglioneuroma-paraganglioma of the intradural filum terminale. Case report. J Neurosurg 1972;36:652-8. 
[PUBMED] |
| 5. | Walsh JC, O'Brien DF, Kumar R, Rawluk D. Paraganglioma of the cauda equina: A case report and literature review. Surgeon 2005;3:113-6. 
[PUBMED] |
| 6. | Pacak K, Linehan WM, Eisenhofer G, Walther MM, Goldstein DS. Recent advances in genetics, diagnosis, localization, and treatment of pheochromocytoma. Ann Intern Med 2001;134:315-29. 
[PUBMED] |
| 7. | Frewin DB, Jamieson GG, Russell WJ, Chatterton BE, Ropiha C, Boundy KL, et al. Extra-adrenal phaeochromocytoma: Report of three interesting cases. Aust N Z J Surg 1989;59:691-5. 
[PUBMED] |
| 8. | Toyota B, Barr HW, Ramsay D. Hemodynamic activity associated with a paraganglioma of the cauda equina. Case report. J Neurosurg 1993;79:451-5. 
[PUBMED] |
| 9. | Zileli M, Kalayci M, Basdesmir G. Paraganglioma of the thoracic spine. J Clin Neurosci 2008;15:823-7. 
|
| 10. | Lenz T, Gossman J, Schulte KL, Salewski L, Geiger H. Diagnosis of pheochromocytoma. Clin Lab 2002;48:5-18. 
|
| 11. | Krentz AJ, Hale PJ, Horrocks PM, Heslop KE, Johnston DG, Wright AD, et al. Metabolic effects of pharmacological adrenergic blockade in phaeochromocytoma. Clin Endocrinol 2002;50:217-9. 
|
| 12. | Boncoeur-Martel MP, Lesort A, Moreau JJ, Labrousse F, Roche I, Bouillet P, et al. MRI of paraganglioma of the filum terminale. J Comput Assist Tomogr 1996;20:162-5. 
|
| 13. | Levy RA. Paraganglioma of the filum terminale: MR findings. AJR Am J Roentgenol 1993;160:851-2. 
[PUBMED] |
| 14. | Moran CA, Rush W, Mena H. Primary spinal paragangliomas: A clinicopathological and immunohistochemical study of 30 cases. Histopathology 1997;31:167-73. 
[PUBMED] |
| 15. | Aghakhani N, George B, Parker F. Paraganglioma of the cauda equina region-report of two cases and review of the literature. Acta Neurochir 1999;141:81-7. 
[PUBMED] |
| 16. | Yang SY, Jin YJ, Park SH, Jahng TA, Kim HJ, Chung CK, et al. Paragangliomas in the cauda equina region: Clinicopathoradiologic findings in four cases. J Neurooncol 2005;72:49-55. 
|
| 17. | Sonneland PR, Scheithauer BW, LeChago J, Crawford BG, Onfrio BM. Paraganglioma of the cauda equina region. Clinicopathologic study of 31 cases with special reference to immunocytology and ultrastructure. Cancer 1986;58:1720-35. 
|
| 18. | Say CC, Hori J, Spratt J Jr. Chemodectoma with distant metastasis: Case report and review of the literature. Am Surg 1973;39:333-41. 
[PUBMED] |
[Figure 1], [Figure 2], [Figure 3], [Figure 4], [Figure 5]
[Table 1]
|