|
 
 |
|
ORIGINAL ARTICLE |
|
|
|
| Year : 2013 | Volume
: 19
| Issue : 2 | Page : 213-218 |
| |
Angiotensin II type 1 receptor A1166C gene polymorphism and essential hypertension in Calabar and Uyo cities, Nigeria
Mary Esien Kooffreh1, Chiaka Ijeoma Anumudu2, Roseline Duke3, Elza Cletus Okpako1, P Lava Kumar4
1 Department of Genetics and Biotechnology, University of Calabar, Nigeria
2 Department of Zoology, University of Ibadan, Nigeria
3 Department of Opthalmology, University of Calabar Teaching Hospital, Calabar, Nigeria
4 Virology Unit, International Institute of Tropical Agriculture, Ibadan, Nigeria
| Date of Web Publication | 5-Aug-2013 |
Correspondence Address:
Mary Esien Kooffreh
Department of Genetics and Biotechnology, University of Calabar, P.M.B 1115, Calabar
Nigeria
 Source of Support: None, Conflict of Interest: None
DOI: 10.4103/0971-6866.116120

 Abstract Abstract | | |
Objectives: The angiotensin II protein is a vasoconstrictor that exerts most of its influence through the angiotensin II type 1 receptor (AT 1 R). Inconsistent association between the A1166C polymorphism of the AT 1 R gene and hypertension has been reported among various populations but not among the peoples of Calabar and Uyo. This study was designed to determine the frequency of the A1166C polymorphism of the AT 1 R gene and its association with hypertension in a sample population of Calabar and Uyo.
Materials and Methods: A population-based case control design consisting of total of 1224 participants, 612 each of patients and controls were randomly recruited from hypertension clinics and the general population. Genotyping of the A1166C allele of the AT 1 R gene to identify variants was performed using polymerase chain reaction and restriction enzyme digestion. Multiple regressions were applied to test whether the A1166 genotypes were predictors of hypertension.
Results: 99% of the study population had the wild type AA genotype, and 1% was AC heterozygous carriers of the A1166C polymorphism.
Conclusion: The A1166C polymorphism was not a predictor of hypertension in the sample population of Calabar and Uyo.
Keywords: A1166C, hypertension, polymorphism
How to cite this article:
Kooffreh ME, Anumudu CI, Duke R, Okpako EC, Kumar P L. Angiotensin II type 1 receptor A1166C gene polymorphism and essential hypertension in Calabar and Uyo cities, Nigeria. Indian J Hum Genet 2013;19:213-8 |
How to cite this URL:
Kooffreh ME, Anumudu CI, Duke R, Okpako EC, Kumar P L. Angiotensin II type 1 receptor A1166C gene polymorphism and essential hypertension in Calabar and Uyo cities, Nigeria. Indian J Hum Genet [serial online] 2013 [cited 2016 May 24];19:213-8. Available from: http://www.ijhg.com/text.asp?2013/19/2/213/116120 |
 Introduction Introduction | |  |
The angiotensin II protein is a well-known vasoconstrictor that exerts most of its influence through the angiotensin II type 1 receptor (AT1R). AT1R is a membrane bound G protein coupled-receptor that mediates the effects of angiotensin II. [1] The highly polymorphic human AT1R gene is 55 kb long having five exons and four introns, A1166C polymorphism is a single base substitution of adenine for cytosine at position 1166 in the 3 ' untranslated regions of the gene located on chromosome 13. The A allele produces the fragment that lacks the restriction site for the Dde1 enzyme but in the presence of cytosine, there is a restriction site for the enzyme (C allele). The physiological significance of the polymorphism is uncertain because data on the function of the AT1R polymorphism is limited. Thus, the mechanism responsible for the association of hypertension status with A1166C polymorphism has remained largely unknown, and the amino acid sequence of the receptor is not altered. It is however, thought to affect mRNA stability and transcription and is in linkage disequilibrium with some other polymorphism. The A1166C polymorphism is associated with prevalent hypertension, increased aortic stiffness. [4],[5],[6] This polymorphism has also been associated with other diseases such as left ventricular hypertrophy [7] pregnancy induced hypertension; early coronary disease and excessive vasoconstriction. [8],[9],[10] Stankovic et al., [2] reported a significant association between this polymorphism and hypertension in males but not females. The frequency of the C1166 allele was high among hypertensives. [11],[12] Some other studies have also reported a negative association between the A1166C polymorphism and hypertension. [13],[14] These variations were attributed to ethnic differences in the various populations. [14],[15]
There are no published data on these polymorphisms in the ethnic populations in Nigeria. This study was carried out to determine if there is an association between A1166C polymorphism and hypertension in a sample population of Calabar and Uyo cities.
 Materials and Methods Materials and Methods | |  |
Population
A total sample population of 1,224 adult men and women from different ethnic groups was included in this study. Of this number, 612 were patients attending the hypertension clinics in the University of Calabar Teaching Hospital, Calabar, the University of Uyo Teaching Hospital, Uyo. The other 612 were individuals whose blood pressure was below 140/90 mmHg, who were also not taking hypertensive drugs and not below the age of twenty from the same population. These individuals served as the control group.
Inclusion criteria
All patients were individuals whose BP were consistently above 140/90 mmHg or were taking hypertensive medications. Controls were individuals whose BP were consistently below 140/90 mmHg and were not taking hypertensive medications.
Exclusion criteria
Females in the population using oral contraceptives were excluded from the study population.
Collection of samples
Blood was obtained from thumb pricks and blotted onto a filter (Whatman, no 3) paper, allowed to dry at room temperature and preserved in plastic bags prior to DNA extraction.
Ethical approval
Subjects included in the study gave informed consent, and ethical approval for the study was obtained from the joint University of Ibadan/University College Hospital ethical review committee and each of the health establishment concerned-University Teaching Hospital Calabar, University of Uyo Teaching Hospital and the General Hospital, Calabar.
Filter paper extraction of DNA
This was done according to Bereczky et al. [15] Pieces [1],[2] of the filter paper about 5 mm in diameter were cut using a sterile blade for each samples. These pieces were placed in an eppendorf tube, soaked in 65 μl of T.E buffer. The tube was incubated at 50°C for 15 min in a water bath. The pieces were pressed gently at the bottom of the tube several times using a new pipette tip for each sample. The eppendorf tubes were heated again for 15 min at 97°C to elute the DNA. The liquid condensing on the lid and the walls of the tube was removed by a short centrifugation (2-3 s). The DNA extract that is the supernatant was kept at −20°C before use.
The angiotensin 11 type 1 receptor gene requires Polymerase Chain Reaction/Restriction Fragment Length Polymorphism digestion to identify alleles. Genomic DNA (4 μl) was amplified in a 25 μl PCR reaction mix containing Promega flexi green buffer 5 μl, dNTPs 0.5 μl, upstream and downstream oligonucleotide primers 0.5 μl each, magnesium chloride 1.5 μl, 12.88 μl of nuclease-free water and Taq DNA polymerase 0.06 μl. AT 1 R gene: Cycling conditions include an initial denaturation of 94°C for 2 min, followed by 40 cycles of a further denaturation at 94°C for 1 min, annealing at 60°C for 1 min, extension 72°C for 2 min, and a final extension of 72°C for 10 min. [3]
A1166C polymorphism of the angiotensin 11 type 1 receptor primer sequence. [3]
5'-AAT GCT TGT AGC CAA AGT CAC CT-3'
5'-GGC TTT GCT TTG TCT TGT TG-3'
A cocktail of 0.25 μl of the Dde1 enzyme, 1 μl of the 10 × buffer D; 0.1 μl of acetyl BSA and 8.5 μl of sterile water was added to 10 μl of the PCR product. The enzyme digestion was performed in a final volume of 19.85 μl at 37°C for 4 h.
Statistical analysis
The statistical package for social sciences-SPSS for windows ® version 16.0 was used to statistically analyze the data obtained. Genotype frequencies in control and hypertensive groups were compared by Chi-square analysis. Continuous variables were compared between hypertensives and controls by independent t-test. Multiple regressions analysis were also carried out using Systolic Blood Pressure or Diastolic Blood Pressure as dependent variable, then sex, age, and BMI were used as independent variables. P < 0.05 was considered statistically significant.
 Results Results | |  |
PCR/enzyme digestion
AT1R gene polymorphism: The Dde1 enzyme cuts the PCR product into two pieces, 600 bp and 250 bp in the A variant. An additional Dde1 recognition site is created in the C variant at nucleotide 1166 located within the 250 bp fragment. The homozygous CC individual produces three bands (600 bp, 140 bp and 110 bp long). The homozygous AA individual produces two bands (600 bp and 250 bp long). The heterozygous individual produces four bands 600 bp, 250 bp, 140 bp and 110 bp long. [3] The homozygous CC individual was not observed [Figure 1].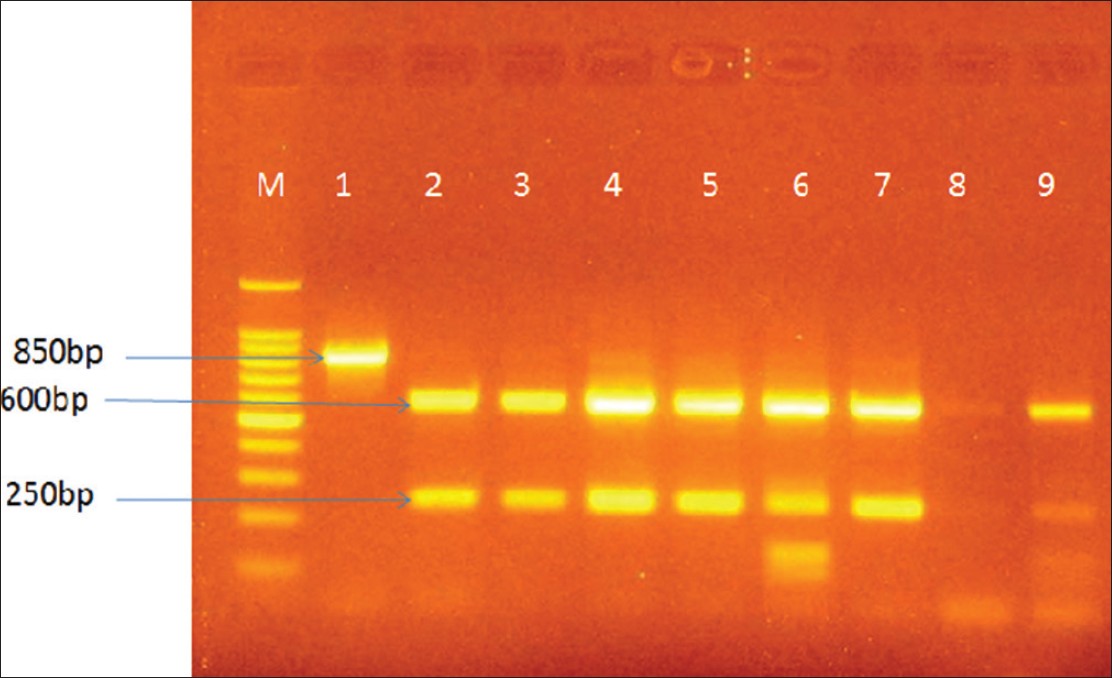 | Figure 1: Agarose gel electrophoresis showing digestion of the 850 bp PCR product by the Dde1 restriction endonuclease. Legend Lane M is the 100 bp DNA ladder Lane 1 is the PCR product Lane 2 is the homozygous AA individual Lane 6 is the heterozygous AC individual
Click here to view |
Genotype and allele distribution
For the A1166C allele, 99% of the study population had the wild type AA genotype and 1% was AC heterozygous carriers of the mutation [Table 1]. The females (55.4%; 64.4%) with the AA genotypes were more than the males (43.6%; 36.4%) in control and patient population respectively [Table 2]. When continuous variables were compared between hypertensive and control groups, significant differences existed between the age, BMI, systolic and diastolic blood pressure of controls and patients. By multiple regression analysis, age was the predictor for SBP and DBP in the hypertensive group r = 0.604 SBP, r = 0.594 DBP, P < 0.05. Age was a predictor for DBP in the control group r = 0.542, gender was a predictor for SBP in the control group. The A1166C gene was not a positive predictor for hypertension in the study population. | Table 1: Genotype and allele frequency distribution among hypertensives and controls
Click here to view |
 | Table 2: Distribution of genotypes by gender among hypertensives and controls
Click here to view |
Body mass index
In the patient population, BMI below 24.9 kg/m 2 was observed in 234 persons (31.54%), BMI between 25 kg/m 2 and 30 kg/m 2 was observed in 193 persons (38.23%) and BMI above 30 kg/m 2 was found in 185 persons (30.23%). In the controls, BMI above 30 kg/m 2 was found in 80 persons (13.07%), BMI between 25 kg/m 2 and 30 kg/m 2 was found in 121 persons (19.77%) and a BMI below 24.9 kg/m 2 was observed in 411 persons (67.16%) [Figure 2]. | Figure 2: Body Mass Index observed among control and patient groups. Legend: Normal ≤24.9 kg/m2 Overweight 25-30 kg/m2 Obese ≥30 kg/m2
Click here to view |
 Discussion Discussion | |  |
The CC allele of the AT1R gene was not observed in this study population. 99% of the population had the AA wild type gene. This is in contrast to what is reported in some literature. A high prevalence of the CC genotype was observed in Chinese hypertensives than controls. [16] In a sample of Swedish twins, Iliadou et al., [17] did not observe any association between the ACE I/D polymorphism or AT1R A1166C polymorphism and blood pressure. Schmidt et al., [18] also did not detect any association between the A1166C allele and hypertension but a decreased prevalence of C allele was observed among hypertensives. Tiret et al. [19] reported a higher prevalence of the C allele among female hypertensives than controls but no such observation among men. A higher prevalence of the CC genotype was observed in Chinese hypertensives than controls. [20]
Generally large interethnic differences in the frequencies of genotype polymorphisms of the RAS exist in different populations. To explain ethnic differences observed in allele frequency in different study populations, Lynch et al. [21] reported that there are differences in linkage disequilibrium for different loci among various populations if such differences exist in regions where these variants are found, and such variants are not causal but are in linkage disequilibrium with the putative locus, this might explain the inconsistency in results across population. Among the Efiks and the Ibibios, the major ethnic group in this study population, the frequency of the A allele was 99.7 and 0.3, for C allele the average C allele frequency reported in the Chinese population was 0.11. In another study, the homozygous A1166 allele frequency was 92.8 among the studied subjects. The frequency of homozygous A allele was significantly higher in the hypertensives than the normotensive subjects (97.5% and 80% respectively) with a higher frequency among male patients. [22]
Farrag et al., [22] proposed that the A allele may be a predisposing factor for essential hypertension in Egyptians. Zhenni et al., [23] observed only two genotypes AA and AC of the AT1IR polymorphism, but reported a higher frequency of the A allele among the patients than the controls. Lee and Kim, [24] observed 96% and 6% for A and C allele of the angiotensin 11 type 1 receptor polymorphism in Korea. Hahntow et al. [25] reported the A allele to be associated with high blood pressure. He also reported that the A allele showing an association was not totally out of place given the fact that this locus had no major impact on hypertension phenotype as suggested by genome wide studies. [26],[27],[28] Other studies [2],[17] have reported the C allele instead to be associated with hypertension.
Most patients were married and advanced in age (above 40 years). Increase in age is thought to increase blood pressure because the arteries become hardened, less active, kidney function decreases and the body does not process salt as well as before. [29] It was observed that most individuals below the age of 40 years were normotensives.
Most controls in the study had a BMI less than or equal to 24.9 kg/m 2 (67.10%). In the patient population, overweight 38.23% and obesity 30.23% reach a moderate prevalence among patients. Hypertension has been reported to be strongly correlated with BMI. Weight gain in adulthood is seen as an important risk factor for hypertension. [30] Humayun et al., [31] observed a high trend of hypertension with increasing BMI among Pakistanis, with a high incidence among females whose weight was above normal that is more than 24.9 kg/m 2 . Positive associations between body mass index and blood pressure have also been documented in cross sectional studies in different Asian population. Ethnic differences existed in the association between BMI and hypertension and optimal mi cutoffs for overweight Chinese, Indonesians and Vietnamese adults. [32],[33],[34]
 Conclusion Conclusion | |  |
A1166C polymorphism is not associated with hypertension in the study population of Calabar and Uyo.
 References References | |  |
| 1. | de Gasparo M, Catt KJ, Inagami T, Wright JW, Unger T. International union of pharmacology. XXIII. The angiotensin II receptors. Pharmacol Rev 2000;52:415-72. 
|
| 2. | Stankoviæ A, Zivkovic M, Glisiæ S, Alavantiæ D. Angiotensin II type 1 receptor gene polymorphism and essential hypertension in Serbian population. Clin Chim Acta 2003;327:181-5. 
|
| 3. | Lapierre AV, Arce ME, Lopez JR, Ciuffo GM. Angiotensin II type 1 receptor A1166C gene polymorphism and essential hypertension in San Luis. Biocell 2006;30:447-55. 
|
| 4. | Benetos A, Gautier S, Ricard S, Topouchian J, Asmar R, Poirier O, et al. Influence of angiotensin-converting enzyme and angiotensin II type 1 receptor gene polymorphisms on aortic stiffness in normotensive and hypertensive patients. Circulation 1996;94:698-703. 
|
| 5. | Wang WY, Zee RY, Morris BJ. Association of angiotensin II type 1 receptor gene polymorphism with essential hypertension. Clin Genet 1997;51:31-4. 
|
| 6. | Danser AH, Schunkert H. Renin-angiotensin system gene polymorphisms: Potential mechanisms for their association with cardiovascular diseases. Eur J Pharmacol 2000;410:303-316. 
|
| 7. | Takami S, Katsuya T, Rakugi H, Sato N, Nakata Y, Kamitani A, et al. Angiotensin II type 1 receptor gene polymorphism is associated with increase of left ventricular mass but not with hypertension. Am J Hypertens 1998;11:316-21. 
|
| 8. | Alvarez R, Reguero JR, Batalla A, Iglesias-Cubero G, Cortina A, Alvarez V, et al. Angiotensin-converting enzyme and angiotensin II receptor 1 polymorphisms: Association with early coronary disease. Cardiovasc Res 1998;40:375-9. 
|
| 9. | van Geel PP, Pinto YM, Buikema H, van Gilst WH. Is the A1166C polymorphism of the angiotensin II type 1 receptor involved in cardiovascular disease? Eur Heart J 1998;19:G13-7. 
|
| 10. | van Geel PP, Pinto YM, Voors AA, Buikema H, Oosterga M, Crijns HJ, et al. Angiotensin II type 1 receptor A1166C gene polymorphism is associated with an increased response to angiotensin II in human arteries. Hypertension 2000;35:717-21. 
|
| 11. | Rubattu S, Di Angelantonio E, Stanzione R, Zanda B, Evangelista A, Pirisi A, et al. Gene polymorphisms of the renin-angiotensin-aldosterone system and the risk of ischemic stroke: A role of the A1166C/AT1 gene variant. J Hypertens 2004;22:2129-34. 
|
| 12. | Dzida G, Sobstyl J, Puzniak A, Golon P, Mosiewicz J, Hanzlik J. Polymorphisms of angiotensin-converting enzyme and angiotensin II receptor type 1 genes in essential hypertension in a Polish population. Med Sci Monit 2001;7:1236-41. 
|
| 13. | Kikuya M, Sugimoto K, Katsuya T, Suzuki M, Sato T, Funahashi J, et al. A/C1166 gene polymorphism of the angiotensin II type 1 receptor (AT1) and ambulatory blood pressure: The Ohasama Study. Hypertens Res 2003;26:141-5. 
|
| 14. | Agachan B, Isbir T, Yilmaz H, Akoglu E. Angiotensin converting enzyme I/D, angiotensinogen T174M-M235T and angiotensin II type 1 receptor A1166C gene polymorphisms in Turkish hypertensive patients. Exp Mol Med 2003;35:545-9. 
|
| 15. | Bereczky S, Mårtensson A, Gil JP, Färnert A. Short report: Rapid DNA extraction from archive blood spots on filter paper for genotyping of Plasmodium falciparum. Am J Trop Med Hyg 2005;72:249-51. 
|
| 16. | Ono K, Mannami T, Baba S, Yasui N, Ogihara T, Iwai N. Lack of association between angiotensin II type 1 receptor gene polymorphism and hypertension in Japanese. Hypertens Res 2003;26:131-4. 
|
| 17. | Iliadou A, Lichtenstein P, Morgenstern R, Forsberg L, Svensson R, de Faire U, et al. Repeated blood pressure measurements in a sample of Swedish twins: Heritabilities and associations with polymorphisms in the renin-angiotensin-aldosterone system. J Hypertens 2002;20:1543-50. 
|
| 18. | Schmidt S, Beige J, Walla-Friedel M, Michel MC, Sharma AM, Ritz E. A polymorphism in the gene for the angiotensin II type 1 receptor is not associated with hypertension. J Hypertens 1997;15:1385-8. 
|
| 19. | Tiret L, Blanc H, Ruidavets JB, Arveiler D, Luc G, Jeunemaitre X, et al. Gene polymorphisms of the renin-angiotensin system in relation to hypertension and parental history of myocardial infarction and stroke: The PEGASE study. Projet d'Etude des Gènes de l'Hypertension Artérielle Sévère à modérée Essentielle. J Hypertens 1998;16:37-44. 
|
| 20. | Jiang Z, Zhao W, Yu F, Xu G. Association of angiotensin II type 1 receptor gene polymorphism with essential hypertension. Chin Med J (Engl) 2001;114:1249-51. 
|
| 21. | Lynch AI, Boerwinkle E, Davis BR, Ford CE, Eckfeldt JH, Leiendecker-Foster C, et al. Pharmacogenetic association of the NPPA T2238C genetic variant with cardiovascular disease outcomes in patients with hypertension. JAMA 2008;299:296-307. 
|
| 22. | Farrag W, Eid M, El-Shazly S, Abdallah M. Angiotensin II type 1 receptor gene polymorphism and telomere shortening in essential hypertension. Mol Cell Biochem 2011;351:13-8. 
|
| 23. | Zhenni J, Wensheng Z, Feng Y, Geng X Association of angiotensin II type 1 receptor gene polymorphism with essential hypertension Chin Med J 2001; 114(12):1249-1251 
|
| 24. | Lee KB, Kim UK. Angiotensinogen and angiotensin II type 1 receptor gene polymorphism in patients with autosomal dominant polycystic kidney disease: Effect on hypertension and ESRD. Yonsei Med J 2003;44:641-7. 
|
| 25. | Hahntow IN, Mairuhu G, van Valkengoed IG, Koopmans RP, Michel MC. Are functionally related polymorphisms of renin-angiotensin-aldosterone system gene polymorphisms associated with hypertension. BMC Cardiovasc Disord 2010;10:23. 
|
| 26. | Wu X, Kan D, Province M, Quertermous T, Rao DC, Chang C, et al. An updated meta-analysis of genome scans for hypertension and blood pressure in the NHLBI Family Blood Pressure Program (FBPP). Am J Hypertens 2006;19:122-7. 
|
| 27. | Rice T, Cooper RS, Wu X, Bouchard C, Rankinen T, Rao DC, et al. Meta-analysis of genome-wide scans for blood pressure in African American and Nigerian samples. The National Heart, Lung, and Blood Institute GeneLink Project. Am J Hypertens 2006;19:270-4. 
|
| 28. | Newton-Cheh C, Johnson T, Gateva V, Tobin MD, Bochud M, Coin L, et al. Genome-wide association study identifies eight loci associated with blood pressure. Nat Genet 2009;41:666-76. 
|
| 29. | Lloyd-Jones DM, Evans JC, Levy D. Hypertension in adults across the age spectrum: Current outcomes and control in the community. JAMA 2005;294:466-72. 
|
| 30. | Jafar TH, Chaturvedi N, Pappas G. Prevalence of overweight and obesity and their association with hypertension and diabetes mellitus in an Indo-Asian population. CMAJ 2006;175:1071-7. 
|
| 31. | Humayun A, Shah AS, Sultana R. Relation of hypertension with body mass index and age in male and female population of Peshawar, Pakistan. J Ayub Med Coll Abbottabad 2009;21:63-5. 
|
| 32. | Stevens J, Truesdale KP, Katz EG, Cai J. Impact of body mass index on incident hypertension and diabetes in Chinese Asians, American Whites, and American Blacks: The People's Republic of China Study and the Atherosclerosis Risk in Communities Study. Am J Epidemiol 2008;167:1365-74. 
|
| 33. | Colin Bell A, Adair LS, Popkin BM. Ethnic differences in the association between body mass index and hypertension. Am J Epidemiol 2002;155:346-53. 
|
| 34. | Nguyen TT, Adair LS, Suchindran CM, He K, Popkin BM. The association between body mass index and hypertension is different between East and Southeast Asians. Am J Clin Nutr 2009;89:1905-12. 
|
[Figure 1], [Figure 2]
[Table 1], [Table 2]
|